Direct vs. Flux-Assisted Solid-State Synthesis: A Comparative Guide for Advanced Material and Drug Development
This article provides a comprehensive comparison between direct solid-state reaction and flux-assisted synthesis methods, tailored for researchers and professionals in material science and drug development.
Direct vs. Flux-Assisted Solid-State Synthesis: A Comparative Guide for Advanced Material and Drug Development
Abstract
This article provides a comprehensive comparison between direct solid-state reaction and flux-assisted synthesis methods, tailored for researchers and professionals in material science and drug development. It explores the fundamental principles of each technique, detailing their specific methodological applications in creating functional materials like oxysulfide photocatalysts and battery components. The scope extends to troubleshooting common challenges such as impurity control and particle size management, and concludes with rigorous validation and comparative analysis frameworks. By synthesizing current research, this guide aims to equip scientists with the knowledge to select and optimize synthesis pathways for enhanced material performance and scalability.
Understanding Solid-State Synthesis: Core Principles of Direct and Flux-Assisted Methods
Direct solid-state reaction is a foundational synthetic method in inorganic and solid-state chemistry for producing polycrystalline materials from solid starting reagents. This method involves the direct reaction of solid precursors at elevated temperatures to form a new chemical compound, without the involvement of solvents or liquid intermediates [1]. The process is characterized by its reliance on high-temperature heating to facilitate atomic diffusion across particle boundaries, leading to nucleation and crystal growth of the desired product phase [1] [2]. The fundamental steps typically include initial contact between reactant particles, chemical reaction at the interface, formation of a new product layer, and subsequent crystal growth, often accompanied by repeated grinding and heating to improve product homogeneity and yield [2].
The widespread adoption of direct solid-state reactions stems from several inherent advantages, particularly for large-scale industrial production. These advantages include procedural simplicity, the absence of solvents, and the capability for high-throughput synthesis of highly crystalline materials with minimal defects [1] [2]. The method reliably produces thermodynamically stable phases, making it particularly suitable for manufacturing ceramic oxides, phosphors, battery electrode materials, and high-temperature superconductors [1] [3]. However, these benefits are counterbalanced by significant limitations, including poor control over final particle morphology, irregular crystal sizes and shapes, difficulty in achieving uniform reagent mixing, and the frequent necessity for extremely high sintering temperatures that can exceed 1400°C [1] [2] [3]. Furthermore, the method often fails to yield metastable phases, as the high-temperature conditions inherently favor the most thermodynamically stable products [2].
Fundamental Mechanisms and Reaction Kinetics
The mechanism of direct solid-state reactions is governed by a complex interplay of thermodynamics and kinetics. From a thermodynamic perspective, the synthesis process can be visualized as navigating a material's energy landscape, where the system moves from a mixture of stable precursor phases into a new free energy basin representing the target material [2]. The primary driving force for these reactions is the change in Gibbs free energy (ΔG), with more negative values generally favoring faster reaction rates [4]. However, several kinetic barriers must be overcome for successful product formation.
The reaction process initiates with contact between particles of the solid reactants, followed by a chemical reaction at their surfaces. This leads to the formation of an initial product layer that subsequently separates the reacting phases [2]. Further reaction progress depends critically on solid-state diffusion, where atoms or ions migrate through the product layer to continue the reaction at the interface [1] [2]. These diffusion processes require overcoming significant activation energies and are strongly influenced by morphological factors such as reactant surface area, particle size, and the intimacy of reagent mixing [1].
Table 1: Key Factors Influencing Solid-State Reaction Mechanisms
| Factor Category | Specific Factors | Impact on Reaction Mechanism |
|---|---|---|
| Chemical Properties | Reactant reactivity, Free energy change (ΔG) | Determines thermodynamic driving force and reaction feasibility [1] |
| Morphological Properties | Reactant surface area, Particle size distribution | Influences diffusion distances and contact areas between reagents [1] |
| Reaction Conditions | Temperature, Pressure, Atmosphere, Heating time | Affects diffusion coefficients and reaction rates; can prevent sublimation or control reaction reversibility [1] |
| Nucleation & Growth | Interfacial energy, Diffusion rates | Controls the initial formation and subsequent development of the product phase [2] |
A significant mechanistic challenge in solid-state synthesis is the formation of stable intermediates that can consume the available thermodynamic driving force and prevent the target material from forming. These intermediates, often highly stable byproducts, can effectively sequester reactants and create kinetic barriers that inhibit the desired reaction pathway [4]. This phenomenon is particularly problematic when synthesizing metastable materials, which represent countless functional technologies but require careful kinetic control to avoid the formation of more thermodynamically favorable competing phases [4].
Diagram 1: Competitive pathways in solid-state reaction. The target material forms only when ΔG₂ is favorable over ΔG₃.
Experimental Protocols in Direct Solid-State Synthesis
Traditional Solid-State Reaction Protocol
The conventional methodology for direct solid-state synthesis follows a systematic protocol focused on achieving homogeneous mixing and sufficient thermal energy for reaction completion. The following procedure outlines the general steps, with specific examples provided for context:
- Precursor Preparation: Select high-purity solid precursors (typically oxides, carbonates, or other salts) and weigh them in the appropriate stoichiometric ratios corresponding to the target compound's composition [3].
- Mixing and Grinding: Combine the solid precursors in a mortar and pestle or mechanical mill, and grind thoroughly to achieve intimate mixing and reduce particle size. This step is crucial for increasing contact surfaces between reactants and shortening diffusion paths [3]. An appropriate amount of anhydrous ethanol may be added during grinding to create a slurry that facilitates more uniform mixing, which is subsequently evaporated [3].
- Calcination: Transfer the homogenized powder into a suitable container (e.g., alumina crucible) and heat in a controlled atmosphere furnace (air, oxygen, or inert gas, depending on the material system). The heating profile typically includes a ramp to an intermediate temperature with a sustained hold to initiate reaction and decompose carbonates or other anion species [3].
- Secondary Grinding and Pelletization: After the initial calcination, the reacted powder is removed, ground again to break up aggregates, and often pressed into pellets. Pelletization enhances interparticle contact and reduces porosity, thereby improving solid-state diffusion in subsequent heating steps [2].
- High-Temperature Sintering: Heat the pellets at the final sintering temperature (often significantly higher than the calcination temperature) for an extended period (typically several hours to days) to complete crystallization and achieve phase purity [1] [3].
- Product Characterization: The final product is ground into a powder and characterized using techniques such as X-ray diffraction (XRD) for phase identification, scanning electron microscopy (SEM) for morphological analysis, and various spectroscopic methods to determine functional properties [3].
Key Research Reagent Solutions
Table 2: Essential Materials and Reagents for Solid-State Synthesis Experiments
| Reagent/Equipment | Function/Role in Synthesis | Specific Examples |
|---|---|---|
| Oxide Precursors | Source of metal cations in the target material | MgO, TiO₂, MnO₂ [3] |
| Carbonate Precursors | Source of metal cations; decomposes to release CO₂ | MnCO₃, Li₂CO₃ [3] |
| Flux Agents | Lower synthesis temperature and improve morphology | LiCl, Li₂CO₃ [3] |
| Grinding Media | Achieve homogeneous mixing of solid precursors | Agate mortar and pestle, mechanical mill [3] |
| High-Temperature Crucibles | Withstand extreme sintering temperatures | Alumina (Al₂O₃) crucibles [3] |
| Programmable Furnaces | Provide controlled heating atmospheres and profiles | Muffle furnace with gas flow control [3] |
Performance Comparison: Traditional vs. Flux-Assisted Methods
The limitations of traditional solid-state synthesis have driven the development of modified approaches, with flux-assisted methods representing a significant advancement. The table below provides a quantitative comparison of these methods using experimental data from the synthesis of Mg₂TiO₄:Mn⁴⁺ (MTO:Mn⁴⁺) phosphor, a representative material system.
Table 3: Experimental Comparison of Traditional and Flux-Assisted Solid-State Synthesis [3]
| Parameter | Traditional Solid-State Method | Flux-Assisted Solid-State Method |
|---|---|---|
| Sintering Temperature | 1300-1400°C required | 950°C sufficient with 50 wt% LiCl flux |
| Phase Purity | Mixture of MTO and MgTiO₃ even at 1400°C | Pure MTO phase achieved at 950°C |
| Particle Morphology | Large, irregular particles with uneven sizes | Improved morphology with more uniform particle size |
| Secondary Phases | Persistent MgTiO₃ impurities | Minimal impurities when optimal flux used |
| Process Simplicity | Simple but requires high-temperature equipment | Additional washing step needed to remove flux residues |
| Luminescence Performance | Good performance when phase-pure | Excellent luminescence with broad excitation band and narrow red emission at 662 nm |
The data demonstrates that the incorporation of a flux agent (LiCl in this case) dramatically reduces the required synthesis temperature by approximately 450°C while simultaneously improving phase purity. This temperature reduction directly translates to lower energy consumption and potentially cheaper equipment requirements. The flux functions by forming a liquid phase at the synthesis temperature that enhances mass transport between solid reactants, effectively facilitating atomic diffusion and promoting the formation of the thermodynamically favored product at significantly lower temperatures [3]. Similar benefits have been observed with other flux agents, including Li₂CO₃, though the specific effectiveness depends on the chemical compatibility with the target material system [3].
Diagram 2: Comparison of traditional and flux-assisted synthesis workflows. The flux method is shorter and operates at lower temperatures.
Beyond flux-assisted approaches, other synthetic strategies have been developed to overcome the limitations of traditional solid-state reactions. Synthesis in the fluid phase, including hydrothermal methods, utilizes solvent media to facilitate reagent diffusion and increase reaction rates [2]. In these approaches, convection effects and active stirring within the fluid reaction medium enhance mass transport compared to solid-state diffusion, with nucleation typically representing the rate-limiting step rather than growth [2]. More advanced computer-aided optimization approaches, such as the ARROWS3 algorithm, combine computational thermodynamics with experimental feedback to identify optimal precursor combinations that avoid the formation of stable intermediates that consume the driving force for target phase formation [4].
Emerging Research and Future Perspectives
Recent advancements in solid-state synthesis are increasingly focused on integrating computational guidance and machine learning to overcome traditional limitations. The ARROWS3 algorithm represents a significant innovation by actively learning from experimental outcomes to identify precursor combinations that avoid the formation of highly stable intermediates, thereby retaining sufficient thermodynamic driving force to form the target material [4]. This approach has demonstrated effectiveness in identifying optimal precursor sets for complex materials like YBa₂Cu₃O₆.₅ (YBCO) while requiring substantially fewer experimental iterations compared to black-box optimization methods [4].
The prediction of synthesizability remains a fundamental challenge in solid-state chemistry. While the energy above hull (Eₕᵤₗₗ) metric derived from density functional theory calculations has been widely used as a proxy for synthesizability, it fails to account for kinetic barriers and entropic contributions that significantly impact actual synthesis feasibility [5]. Novel approaches using positive-unlabeled (PU) learning are emerging to predict the solid-state synthesizability of hypothetical compounds by learning from human-curated literature data, offering promising alternatives to purely thermodynamics-based predictions [5].
For specialized applications requiring extremely high crystal quality, flux-regulated crystallization (FRC) methods with feedback control systems have been developed to maintain stable linear growth rates during single crystal growth [6]. When applied to materials like metal halide perovskites, this approach enables the synthesis of centimeter-scale single crystals with exceptional crystallinity, as quantified by a full width at half-maximum of 15.3 arcsec in X-ray rocking curve measurements [6]. Such controlled growth methods represent a significant advancement beyond conventional solid-state reactions for applications where crystal perfection directly influences material performance.
Flux-assisted synthesis is a versatile method for material preparation that utilizes a molten salt or metal to create a high-temperature solvent medium, facilitating the dissolution and subsequent crystallization of solid-state compounds. This technique has emerged as a powerful alternative to conventional synthesis methods, particularly for preparing complex inorganic materials, two-dimensional (2D) nanostructures, and advanced phosphors with enhanced properties. The fundamental principle involves using a flux—a substance that becomes liquid at the reaction temperature—to provide an ionic environment that promotes mass transport, reduces reaction energy barriers, and controls crystal growth kinetics. Molten salts, serving as the preferred flux medium, offer remarkable advantages including reliability, simplicity, scalability, sustainability, low cost, ease of removal, and a wide temperature window for material synthesis [7].
The era of two-dimensional van der Waals materials has accelerated the adoption of flux-assisted methods, as researchers seek reliable pathways to synthesize high-quality nanomaterials with controlled morphology and enhanced physicochemical properties. Since the isolation of graphene in 2004, the materials science community has explored numerous 2D materials including transition metal dichalcogenides (TMDs), transition metal oxides (TMOs), MXenes, transition metal nitrides (TMNs), and various hybrid frameworks [7]. Traditional synthesis approaches, including mechanical exfoliation, chemical vapor deposition (CVD), and wet chemistry methods, often face limitations in production efficiency, substrate requirements, surfactant contamination, and scalability. Flux-assisted synthesis effectively addresses these challenges through its unique reaction environment, establishing itself as an indispensable tool in modern materials chemistry [7] [8].
Fundamental Principles and Mechanisms
Thermodynamic Foundations
The flux method operates on principles derived from classical nucleation theory, where crystal formation occurs through precipitation from a supersaturated liquid solution. The process begins with the flux material melting and dissolving the precursor elements, followed by controlled cooling that induces supersaturation and subsequent crystal nucleation and growth [8]. The thermodynamic driving force can be understood through the supersaturation ratio (S), defined as S = C/Ceq, where C represents the solute concentration and Ceq is the equilibrium concentration that varies with temperature. At high temperatures, S < 1, maintaining all materials in solution. As the temperature decreases during controlled cooling, Ceq decreases while C remains constant, causing S to increase beyond 1 and triggering the nucleation process [8].
The nucleation thermodynamics is governed by the total Gibbs free energy (ΔG) of the nucleating phase, which for a spherical nucleus of radius r can be expressed as ΔG = 4πr²γ + (4/3)πr³Gv, where γ represents the surface energy and Gv denotes the volume energy. The critical radius (rc) at which nucleus growth becomes thermodynamically favorable occurs when dΔG/dr = 0, yielding rc = 2γvm/ΔGv, where vm is the molar volume. The associated activation energy barrier is ΔGc = (4/3)πγrc³. The nucleation rate follows an Arrhenius relationship: dN/dt = A exp(-ΔGc/kBT), where A is a proportionality constant, kB is Boltzmann's constant, and T is temperature [8]. This theoretical framework explains why nucleation is suppressed at high temperatures while becoming favorable during controlled cooling, enabling precise control over crystal size and quality.
Molten Salt Reaction Environment
Molten salts create a unique reaction environment characterized by several advantageous features. They provide a bare ion environment that eliminates the need for ion desolvation required in conventional solution methods, significantly reducing the energy barrier for reactions. The intense polarization effect of molten salts decreases the stability of chemical bonds in precursor materials, thereby lowering the required reaction temperature. Additionally, the non-solvated ions exhibit strong template and structure-guiding effects, effectively adsorbing to high-energy surfaces of growing materials to stabilize them and control morphology [7].
The selection of appropriate salt systems depends on several factors including melting point, viscosity, solubility of precursors, and ease of removal after reaction. Common salt systems include chlorides (KCl-NaCl, LiCl-KCl), nitrates (Ca(NO3)2-NaNO3), carbonates (K2CO3-Na2CO3), and iodides (NaI) [7] [9]. Each system offers distinct advantages for specific applications, with melting points ranging from approximately 200°C for some eutectic mixtures to over 800°C for single-component salts. The wide temperature window accessible through salt selection enables optimization of reaction conditions for diverse material systems [7].
Comparative Analysis: Flux vs. Conventional Methods
Advantages and Limitations
Table 1: Comparison between flux-assisted synthesis and conventional methods for material preparation
| Aspect | Flux-Assisted Synthesis | Conventional Solid-State Reaction | Wet Chemical Methods |
|---|---|---|---|
| Reaction Temperature | Lower due to reduced energy barriers and polarization effects | Higher temperatures required | Moderate to low temperatures |
| Crystal Quality | High crystalline quality with controlled morphology | Variable, often with defects | Often contains surfactants/residues |
| Scalability | Highly scalable for industrial applications | Limited by diffusion constraints | Limited by solvent volume and cost |
| Morphology Control | Excellent control through salt selection and parameters | Limited control over morphology | Good control but with additive contamination |
| Production Efficiency | High yield and efficiency | Low production efficiency | Moderate efficiency |
| Cost Considerations | Low-cost and sustainable | Moderate cost | Can be expensive with solvents |
| Product Purity | High purity with easy salt removal | Potential for impurities | Residual surfactants affect properties |
Flux-assisted synthesis demonstrates distinct advantages over conventional methods across multiple parameters. Compared to mechanical exfoliation, which suffers from low production efficiency, and chemical vapor deposition, which requires substrate removal, flux methods offer a streamlined pathway to high-quality materials [7]. Unlike wet chemistry approaches that often introduce surfactants affecting material properties, flux synthesis minimizes contamination through easy removal of the salt medium after reaction completion [7]. The method particularly excels in producing large, high-quality single crystals—with sizes sometimes reaching up to 1 cm—that are essential for investigating intrinsic physical properties of materials [8].
The limitations of flux-assisted synthesis primarily relate to the specific salt selection process, which often requires empirical optimization, and the potential for residual flux incorporation if not properly removed. Additionally, some salt systems may present corrosion challenges to reaction vessels, particularly at elevated temperatures. Nevertheless, the overall benefits position flux methods as superior for numerous applications, especially where crystal quality, phase purity, and scalability are paramount [7] [8].
Performance Comparison Through Experimental Data
Table 2: Experimental performance comparison of materials synthesized via different methods
| Material | Synthesis Method | Key Performance Metrics | Results | Reference |
|---|---|---|---|---|
| Ba2Bi3Ta2O11Cl | Conventional Solid-State | O2 evolution activity | Baseline (9 μmol h⁻¹) | [10] |
| Ba2Bi3Ta2O11Cl | Molten Salt Treatment | O2 evolution activity | 2× improvement | [10] |
| W-doped Ba2Bi3Ta2O11Cl | Flux-Assisted Synthesis | O2 evolution activity | 55 μmol h⁻¹ (6× improvement) | [10] |
| (Ca,Zn)TiO3:Pr³⁺ | Solid-State without flux | Afterglow intensity & duration | Baseline | [11] |
| (Ca,Zn)TiO3:Pr³⁺ | Flux-Assisted (NH₄F) | Afterglow intensity & duration | Significant enhancement | [11] |
| Sn2TiO4 | Conventional | Photocatalytic H₂ evolution yield | 55% yield | [8] |
| Sn2TiO4 | Flux Method (optimized ratio) | Photocatalytic H₂ evolution yield | 90% yield | [8] |
| RE3Si2SexS8-xI | Flux-Assisted BCM Method | Band gap (eV) | 2.2-2.5 eV | [9] |
Experimental data consistently demonstrates the performance enhancements achievable through flux-assisted synthesis. In photocatalytic applications, tungsten-doped layered perovskite oxychloride prepared via molten salt methods exhibited oxygen evolution activity six times higher than conventionally prepared materials [10]. Similarly, flux-assisted synthesis of Sn2TiO4 increased yield from 55% to 90% compared to conventional approaches [8]. For luminescent materials, flux addition significantly enhanced the afterglow properties of (Ca,Zn)TiO3:Pr³⁺ phosphors, with different fluxes (NH₄F, NH₄Cl, H₃BO₃, NH₄Br, NH₄I, NH₄BF₄) producing varying degrees of improvement in crystallinity, particle morphology, and luminescence efficiency [11].
The reactive flux-assisted boron-chalcogen mixture (BCM) method has proven particularly effective for synthesizing complex mixed-anion compounds, including rare-earth-based chalcogenide iodides RE3Si2SexS8-xI (RE = La, Ce, Pr, Nd), which exhibited tunable band gaps in the 2.2-2.5 eV range, making them promising semiconductor materials [9]. Similarly, diamond-like HgIn2Q4 (Q = Se, Te) compounds prepared via this route displayed noteworthy nonlinear optical properties attributable to their unique tetrahedral motifs [12].
Experimental Protocols and Methodologies
General Flux-Assisted Synthesis Procedure
The fundamental workflow for flux-assisted synthesis involves several standardized steps, though specific parameters vary based on the target material. The process begins with precursor preparation, where high-purity starting materials are precisely weighed in stoichiometric ratios. For oxide-based materials, this typically involves metal oxides, carbonates, or occasionally elemental precursors. The flux material is then added in predetermined proportions, with typical flux-to-precursor ratios ranging from 1:1 to 14:1 depending on the system requirements [8] [9].
The mixture is thoroughly ground using a mortar and pestle or mechanical mill to ensure homogeneous mixing, then transferred to a reaction vessel—typically an alumina crucible, quartz tube, or graphite container depending on the reaction temperature and chemical compatibility. The vessel is sealed under vacuum or inert atmosphere (argon, nitrogen) to prevent oxidation during heating, particularly when using air-sensitive precursors or fluxes [8] [9].
The sealed vessel is placed in a programmable furnace and subjected to a specific thermal profile: heating to the target temperature at a controlled rate (typically 1-5°C/min), dwelling at this temperature for sufficient time to ensure complete reaction and crystal growth (hours to days), followed by controlled cooling (0.1-5°C/min) to promote proper crystallization. Finally, the flux medium is removed through washing with appropriate solvents (water, methanol, etc.), centrifugation, or mechanical separation, yielding the pure product [7] [8] [9].
Specific Protocol Examples
Protocol 1: Rare-Earth Mixed Chalcogenide Iodides RE3Si2SexS8-xI For synthesizing La3Si2Se1.21S6.79I, Ce3Si2Se1.39S6.61I, Pr3Si2Se1.22S6.78I, and Nd3Si2Se1.18S6.82I, researchers employed a reactive flux-assisted boron-chalcogen mixture method. Precursors included RE2O3 (RE = La, Nd), CeO2, or Pr6O11 (50 mg), SiO2 (5 mg), sulfur powder (40 mg), selenium powder (10 mg), boron (20 mg), and NaI flux (250 mg). These materials were combined in heavily carbon-coated fused silica tubes, evacuated to 10⁻⁴ torr, and flame-sealed. The thermal profile consisted of heating to 850°C in 20 hours, dwelling for 20 hours, cooling to 550°C in 20 hours, followed by natural cooling to room temperature. The resulting products demonstrated stability to air and moisture and were washed with methanol to remove residual flux [9].
Protocol 2: Tungsten-Doped Layered Perovskite Oxychloride The synthesis of Ba2Bi3Ta2O11Cl employed a one-step molten salt method with subsequent tungsten doping. The molten salt treatment significantly altered the crystal morphology and structure, enhancing the oxygen evolution activity. W-doping further improved charge separation efficiency and reduced the energy barrier for water oxidation. The optimized W-doped sample after molten salt treatment exhibited O2 production activity of 55 μmol h⁻¹ without cocatalysts, representing a six-fold enhancement compared to pristine Ba2Bi3Ta2O11Cl prepared conventionally [10].
Protocol 3: (Ca,Zn)TiO3:Pr³⁺ Phosphors Red-emitting afterglow phosphors were prepared using flux-assisted high-temperature solid-state reaction. Precursors included Pr6O11, CaCO3, ZnO, and TiO2 in stoichiometric ratios. Various chemical fluxes (NH4Cl, NH4F, NH4I, NH4Br, NH4BF4, H3BO3) were added at 10 mol% concentration. The mixture was thoroughly ground, heated to 1100-1300°C for 2-4 hours in a muffle furnace, then slowly cooled to room temperature. The flux addition significantly enhanced photoluminescence intensity, crystallinity, and afterglow properties, with NH4F proving particularly effective among the tested fluxes [11].
Essential Research Reagents and Materials
Table 3: Key research reagents and their functions in flux-assisted synthesis
| Reagent Category | Specific Examples | Function in Synthesis | Application Examples |
|---|---|---|---|
| Alkali Halide Fluxes | NaCl, KCl, NaI, LiCl | Lower melting points, provide ionic environment, enhance diffusion | RE3Si2SexS8-xI [9], Sn2TiO4 [8] |
| Alkaline Earth Halides | CaCl2, SrCl2 | Higher temperature flux options, specific crystal structure direction | Perovskite-type materials |
| Mixed Eutectic Systems | KCl-NaCl, LiCl-KCl, Ca(NO3)2-NaNO3 | Lower melting temperatures through eutectic formation | General 2D materials [7] |
| Metal Fluxes | Ga, In, Sn, Pb, Al, Na | Low melting points, serve as reactive medium or self-flux | Clathrate phases, intermetallics [8] |
| Ammonium Halides | NH4F, NH4Cl, NH4Br, NH4I | Low decomposition temperatures, introduce halides, mineralizer | (Ca,Zn)TiO3:Pr³⁺ phosphors [11] |
| Oxide Fluxes | B2O3, H3BO3, PbO | Fluxing agents for oxide materials, often reactive | Phosphor synthesis [11] |
| Precursor Materials | Metal oxides, carbonates, elemental powders | Source of target compound cations/anions | All flux-assisted syntheses |
| Container Materials | Alumina crucibles, quartz tubes, graphite | Withstand high temperatures, chemically inert | High-temperature reactions |
The selection of appropriate flux materials represents a critical decision point in experimental design. Key considerations include melting point, solubility parameters, chemical compatibility with precursors, volatility at reaction temperature, and ease of removal after reaction. For example, NaI flux has proven effective for incorporating iodide anions into crystal structures, as demonstrated in rare-earth chalcogenide iodides [9]. Similarly, tin(II) chloride serves both as flux and reactant in the synthesis of SnNb2O6 and Sn2TiO4 compounds [8]. The wide variety of available fluxes enables researchers to tailor the reaction environment for specific material systems and desired properties.
Current Challenges and Future Perspectives
Despite significant advancements, flux-assisted synthesis faces several challenges that represent opportunities for future research. The selection of optimal flux systems remains largely empirical, with limited theoretical guidance for matching specific flux properties to target materials. Developing predictive models for flux selection would substantially accelerate materials discovery and optimization [7]. Additionally, understanding the fundamental growth mechanisms of two-dimensional materials in molten salts requires further investigation, though researchers have begun developing reasonable models to describe these processes [7].
Future development directions include expanding the library of compatible flux systems, particularly environmentally benign and recyclable options; enhancing control over crystal size and morphology through advanced cooling protocols and flux mixtures; and integrating flux methods with other synthesis techniques to leverage complementary advantages. The exploration of flux-assisted synthesis for emerging material classes—such as multiferroics, topologically protected materials, and quantum spin liquid candidates—represents a promising frontier [7] [8].
Scaling flux-assisted methods for industrial applications while maintaining precise control over material properties presents both a challenge and opportunity. The inherently scalable nature of flux methods positions them favorably for technology transfer, though optimization of parameters for large-scale reactions requires additional research. As materials demands continue evolving toward more complex, multifunctional systems, flux-assisted synthesis will likely play an increasingly important role in meeting these challenges through its unique combination of versatility, efficiency, and control [7].
Key Differences in Mass Transfer and Reaction Kinetics
In solid-state materials synthesis, two principal methodologies dominate: the direct solid-state reaction method and the flux method. The direct solid-state approach involves heating solid reactant powders together at high temperatures, where reactions proceed primarily through solid-state diffusion across particle boundaries. In contrast, the flux method utilizes a solvent medium (the flux) that dissolves reactant materials at high temperatures, facilitating reaction and crystal growth from the molten state before separation upon cooling. The fundamental distinction between these techniques lies in their dominant mass transport mechanisms and consequent reaction kinetics, which ultimately dictate the synthesis outcomes, including crystal quality, particle morphology, and phase purity. Understanding these differences is crucial for researchers selecting appropriate synthesis strategies for specific material systems and applications, from quantum materials to phosphors and beyond.
Table 1: Fundamental Characteristics of Synthesis Methods
| Feature | Direct Solid-State Reaction | Flux Method |
|---|---|---|
| Primary Mass Transfer Mechanism | Solid-state diffusion through crystal lattices and along grain boundaries | Liquid-phase diffusion and convection in molten flux |
| Typical Reaction Temperature | High (often >1000°C) | Variable, but often lower than solid-state due to flux melting point depression |
| Reaction Environment | Solid-solid interface | Liquid (molten flux) solution |
| Dominant Kinetic Limitations | Nucleation and growth limited by ionic mobility in solids | Solubility, supersaturation, and cooling rate |
| Primary Energy Barrier | Lattice energy and diffusion activation energy | Solvation and crystallization energy |
Mass Transfer Mechanisms: Solid-State Diffusion vs. Liquid-Phase Transport
The mass transfer phenomena governing these two methods operate on fundamentally different physical principles, leading to distinct kinetic profiles and synthesis outcomes.
Mass Transfer in Direct Solid-State Reactions
In direct solid-state synthesis, mass transfer occurs primarily through solid-state diffusion, a process several orders of magnitude slower than liquid-phase transport. This diffusion involves the movement of ions through crystal lattices or along grain boundaries and defects, with kinetics described by the Arrhenius equation and strongly dependent on temperature and activation energy barriers [13]. The process is inherently limited by the need for reactants to overcome significant lattice energy barriers, with diffusion coefficients typically ranging from 10⁻¹⁰ to 10⁻¹⁶ cm²/s in oxide systems at moderate temperatures [14].
The product layer model often describes the progressive formation of a reaction product at the interface between reactant particles. As this layer thickens, it creates increasing resistance to further diffusion, potentially leading to incomplete reactions and residual reactants [15]. This model highlights three potential rate-limiting steps: surface reaction, film diffusion, or diffusion through the product layer, with the latter often dominating in solid-state reactions [15]. Furthermore, real powder systems deviate significantly from ideal geometries, with particle surfaces exhibiting complex "moon landscape" topographies featuring cracks and craters that further complicate diffusion pathways [15].
Mass Transfer in Flux-Mediated Reactions
The flux method fundamentally alters mass transfer by creating a liquid medium that dissolves reactant materials, replacing slow solid-state diffusion with rapid liquid-phase transport. In this environment, dissolved species diffuse through the molten flux with diffusion coefficients typically ranging from 10⁻⁵ to 10⁻⁶ cm²/s—several orders of magnitude faster than solid-state diffusion [16]. This enhanced mobility dramatically increases reaction rates and allows for more homogeneous mixing of reactants at the molecular level.
The flux not only provides a transport medium but also influences reaction kinetics through wetting behavior and convective flows. As the system cools, supersaturation drives nucleation and crystal growth, with mass transfer governing the supply of nutrients to growing crystal surfaces [16]. The choice of flux composition directly impacts these processes; for example, in the growth of NdTa₇O₁₉ crystals, the K₂Mo₃O₁₀–B₂O₃ flux was selected specifically for its high dissolution capability, relatively low melting temperature, and appropriate viscosity characteristics that facilitate effective mass transfer without excessive volatility [16].
Reaction Kinetics: Comparative Analysis of Rate Processes
The kinetic profiles of direct solid-state and flux-mediated reactions reveal fundamental differences in their rate-determining steps and temporal evolution.
Kinetics of Solid-State Reactions
Solid-state reaction kinetics typically follow complex models that account for nucleation and growth processes, often described by the Johnson-Mehl-Avrami-Kolmogorov (JMAK) model or shrinking core models depending on the reaction mechanism. These reactions exhibit characteristically slow initial rates as nucleation establishes reaction interfaces, followed by acceleration during the growth phase, and finally diffusion-limited deceleration as product layers thicken [15]. The temperature dependence of these reactions is pronounced, with effective rate constants following Arrhenius behavior with high activation energies (often 150-400 kJ/mol) corresponding to the energy required for lattice diffusion [13].
The non-ideal nature of real particle systems further complicates kinetic analysis. Surface roughness, particle size distributions, and the development of complex microstructures create deviations from idealized kinetic models. For instance, surface roughness can effectively increase the apparent reaction order with respect to the solid reactant by creating additional reactive surface area and complex diffusion pathways [15]. These factors make complete conversion difficult to achieve in practical timeframes, often necessitating repeated grinding and heat treatments to improve product homogeneity.
Kinetics of Flux-Mediated Reactions
Flux-mediated reactions operate through solution-precipitation mechanisms with fundamentally different kinetic profiles. The process initiates with the dissolution of reactants into the molten flux, followed by reaction between dissolved species, and concludes with nucleation and growth of the product phase as the system becomes supersaturated during cooling [16]. The rate-determining step can shift between these stages depending on specific conditions, but the liquid medium generally enables more rapid kinetics than solid-state alternatives.
The flux environment often allows products to form at significantly lower temperatures than solid-state reactions, reducing thermal energy requirements and potentially stabilizing metastable phases inaccessible through direct solid-state synthesis. For example, in the synthesis of ZnAl₂O₄:Cr³⁺ nanophosphors, the flux method enhanced photoluminescence intensity by 3.82 times compared to conventional methods, demonstrating the kinetic advantages of the flux approach for achieving specific material properties [17]. The cooling rate represents a critical kinetic parameter, with slower cooling (0.5-2°C/h) often employed during initial crystal growth stages to control nucleation density, followed by more rapid cooling (10-50°C/h) once crystals are established [16].
Table 2: Comparative Kinetic Parameters and Outcomes
| Parameter | Direct Solid-State Reaction | Flux Method |
|---|---|---|
| Typical Activation Energy | High (150-400 kJ/mol) | Lower (varies with system) |
| Time to Completion | Hours to days with intermediate grinding | Several hours to days (single cycle) |
| Product Crystallinity | Polycrystalline powders | Often single crystals |
| Common Impurities | Unreacted starting materials, intermediate phases | Flux inclusions, solvent residues |
| Phase Selectivity | Thermodynamically most stable phase | Possible metastable phase access |
| Morphology Control | Limited, depends on starting powders | Enhanced, often faceted crystals |
Experimental Protocols and Methodologies
Direct Solid-State Reaction Protocol
The standard protocol for direct solid-state synthesis involves several critical steps that significantly influence mass transfer and reaction kinetics:
Precursor Preparation: High-purity oxide or carbonate powders (e.g., Nd₂O₃, Ta₂O₅) are precisely weighed in stoichiometric ratios. Pre-annealing of precursors at 1000°C for 24 hours may be performed to remove volatile impurities and ensure phase stability [16].
Mechanical Mixing: Reactants are homogenized through grinding in an agate mortar or ball milling to maximize interfacial contact area. Particle size reduction during this stage directly enhances mass transfer by shortening diffusion pathways.
Calcination: The mixed powders are pressed into pellets (at ~50 kN force for 10 mm diameter pellets) to improve interparticle contact and heated in controlled atmosphere furnaces. Multiple synthesis cycles (5-9) at temperatures between 900-1200°C with intermediate regrinding are typically required to achieve homogeneous products [16].
Characterization: Phase purity is verified by powder X-ray diffraction, with structural and morphological analysis performed using scanning electron microscopy. The product is often obtained as a polycrystalline powder with potential for incomplete reaction and compositional heterogeneity.
Flux Growth Experimental Protocol
The flux method follows a distinct experimental pathway optimized for crystal growth:
Charge Preparation: Polycrystalline precursor (pre-synthesized via solid-state method) is mixed with flux material (e.g., K₂Mo₃O₁₀-B₂O₃) in specific mass ratios (typically 5:1 to 15:1 flux-to-material ratio) and homogenized in an agate mortar [16].
Crucible Loading: The mixture is placed in platinum crucibles (5-10 mL volume), which are then positioned within larger alumina crucibles with covers to minimize flux volatility and contamination.
Thermal Profile: The loaded crucibles undergo a precisely controlled temperature program:
- Heating to a dwell temperature (700-1200°C, typically 1100°C) at 100-300°C/h
- Soaking at the maximum temperature for 2-48 hours (typically 24 hours) to ensure complete dissolution
- Slow cooling (0.5-2°C/h) through the crystallization temperature range
- Rapid cooling (10-50°C/h) to room temperature once crystallization is complete [16]
Product Recovery: The flux matrix is dissolved using appropriate solvents (e.g., deionized water, often with ultrasonic assistance), liberating the well-formed crystals for characterization and application.
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful implementation of these synthesis methods requires specific materials and reagents with carefully considered functions:
Table 3: Essential Research Reagents and Materials
| Reagent/Material | Function | Application Examples |
|---|---|---|
| High-Purity Oxides (Nd₂O₃, Ta₂O₅, etc.) | Primary reactants providing metal cations | Solid-state synthesis of NdTa₇O₁₉, starting materials for flux growth [16] |
| Flux Materials (K₂Mo₃O₁₀, B₂O₃, NH₄Cl) | Solvent medium for dissolution and crystal growth; lowers synthesis temperature | K₂Mo₃O₁₀-B₂O₃ for NdTa₇O₁₉ growth; NH₄Cl for ZnAl₂O₄:Cr³⁺ phosphors [16] [17] |
| Platinum Crucibles | Inert containers resistant to corrosive fluxes at high temperatures | Containment of flux reactions up to 1200°C [16] |
| Alumina Crucibles and Covers | Secondary containment to minimize flux volatility and contamination | Outer protection for platinum crucibles during flux growth [16] |
| Agate Mortars and Pestles | Homogeneous mixing and particle size reduction | Grinding of reactants for solid-state reactions; mixing of precursors with flux [16] |
| Carbon Dots (CDs) | Luminescence enhancers and morphology modifiers | Surface modification of ZnAl₂O₄:Cr³⁺ nanophosphors [17] |
Performance Comparison: Synthesis Outcomes and Material Properties
The distinct mass transfer and kinetic mechanisms in these methods yield dramatically different synthesis outcomes:
Product Characteristics
Direct solid-state reactions typically produce polycrystalline powders with particle sizes and morphologies largely determined by the starting materials and grinding processes. The products often contain structural defects, unreacted phases, and compositional heterogeneity due to diffusion limitations [15]. In contrast, flux methods can yield well-faceted single crystals with sizes up to several millimeters, as demonstrated by the successful growth of NdTa₇O₁₉ crystals reaching 3.5 mm in lateral dimension and 2 mm thickness—the largest reported for this material [16]. These single crystals enable detailed structural characterization and physical property measurements that are challenging with polycrystalline materials.
Functional Performance Differences
The structural perfection achieved through flux growth often translates to enhanced functional properties. For ZnAl₂O₄:Cr³⁺ nanophosphors, the flux method with NH₄Cl enhanced photoluminescence intensity by 3.82 times compared to conventional methods, while carbon dot modification further increased performance with 11.23-fold enhancement [17]. Similarly, single crystals of quantum spin liquid candidates like NdTa₇O₁₉ enable precise determination of magnetic anisotropy essential for understanding exotic magnetic ground states [16]. For optical applications, the improved crystalline perfection and reduced defect concentrations in flux-grown materials typically yield superior performance compared to solid-state derived counterparts.
Limitations and Challenges
Each method presents distinct limitations: solid-state reactions struggle with incomplete conversion, limited morphology control, and high energy requirements, while flux methods face challenges with flux inclusion in crystals, potential contamination, and more complex purification requirements. The flux selection process remains partially empirical, with optimal compositions often identified through systematic screening rather than fundamental principles [17] [16].
Table 4: Comprehensive Method Comparison for Specific Material Systems
| Material System | Synthesis Method | Key Parameters | Performance Outcomes |
|---|---|---|---|
| NdTa₇O₁₉ | Flux (K₂Mo₃O₁₀-B₂O₃) | Flux:material=10:1, 1100°C soak, 1°C/h cooling | 3.5 mm single crystals suitable for magnetic measurements [16] |
| ZnAl₂O₄:Cr³⁺ | Solid-state with NH₄Cl flux | 7% Cr doping, NH₄Cl flux addition | 3.82× PL enhancement compared to non-flux method [17] |
| ZnAl₂O₄:Cr³⁺ | Carbon dot modification | 5wt% CDs addition | 11.23× PL enhancement, improved color purity [17] |
| Rare-earth Heptatantalates | Optimized flux growth | Various RE elements, modified cooling profiles | Single crystals enabling magnetic anisotropy determination [16] |
The choice between direct solid-state and flux-mediated synthesis represents a fundamental strategic decision in materials research, with significant implications for research outcomes. Solid-state methods offer simplicity and directness for powder production but face inherent limitations in mass transfer and kinetic barriers. Flux methods provide enhanced mass transfer and superior crystal quality at the cost of additional process complexity. Recent advances in flux composition optimization and the integration of modifiers like carbon dots continue to expand the capabilities of flux-mediated synthesis, enabling unprecedented control over material properties and performance characteristics.
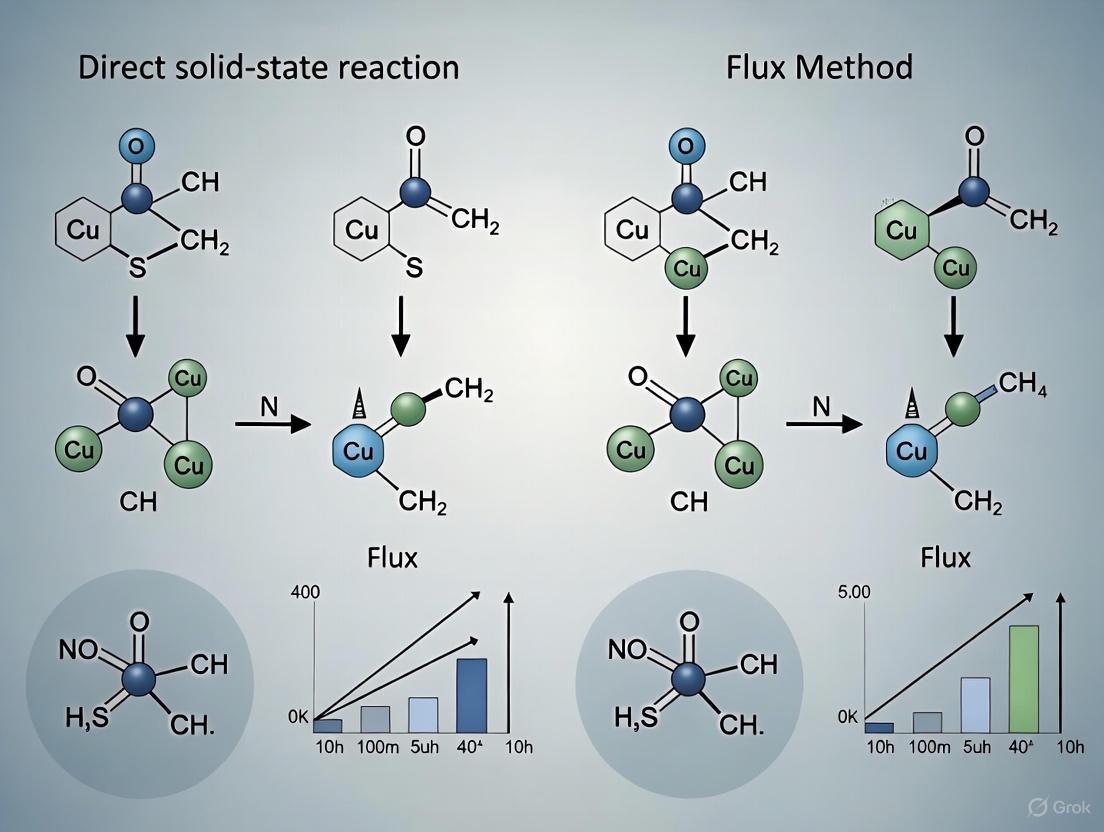
The solid-state reaction method is a cornerstone technique in inorganic materials synthesis, pivotal for producing a wide array of compounds from ceramics to phosphors. The successful formation of target materials with desired phase purity and functional properties is governed by the precise control of three critical process parameters: temperature, time, and atmosphere. Alongside these parameters, the use of flux agents has emerged as a powerful strategy to modify reaction kinetics and thermodynamics, offering a compelling alternative to traditional direct solid-state synthesis. This guide provides an objective comparison between direct and flux-assisted solid-state methods, framing the analysis within ongoing research to achieve superior control over material properties. It is designed to equip researchers and drug development professionals with actionable experimental data and protocols, underscoring the importance of parameter optimization in synthetic pathways.
Theoretical Framework: Thermodynamic and Kinetic Principles
Solid-state reactions are governed by the interplay of thermodynamics and kinetics. The initial phase formed in a reaction often determines the subsequent pathway and final product. Recent research has validated a principle known as the max-ΔG theory, which posits that when the thermodynamic driving force (the change in Gibbs energy, ΔG) to form one product is sufficiently large, thermodynamics dictates the initial product, bypassing kinetic limitations [18]. Analysis of the Materials Project data suggests that approximately 15% of all possible reactions fall within this regime of thermodynamic control [18].
A quantitative study involving in situ characterization of 37 reactant pairs established a threshold for this thermodynamic control. The initial product formation can be predicted when its driving force exceeds that of all other competing phases by ≥60 meV/atom [18]. When multiple phases have a comparable driving force, the reaction enters a kinetic control regime, where factors like diffusion and structural templating dominate. This framework is crucial for understanding how fluxes operate; they can alter local reaction environments to favor thermodynamic products or reduce kinetic barriers.
The following diagram illustrates the decision-making process for selecting a synthesis method based on thermodynamic and kinetic considerations:
Comparative Experimental Data: Flux-Assisted vs. Direct Solid-State Synthesis
The impact of flux agents on critical process parameters is best demonstrated through direct experimental comparisons. The table below summarizes quantitative data from recent studies on phosphor synthesis, highlighting the dramatic improvements achievable with flux methods.
Table 1: Quantitative Comparison of Direct vs. Flux-Assisted Solid-State Synthesis
| Material System | Synthesis Method | Flux Agent | Optimal Temperature (°C) | Soaking Time | Key Performance Metric | Result |
|---|---|---|---|---|---|---|
| Mg₂TiO₄:Mn⁴⁺ (MTO:Mn⁴⁺) [3] | Direct Solid-State | None | 1400 | 12 h | Phase Purity | Mixture of MTO + MgTiO₃ |
| Flux-Assisted | LiCl (50 wt%) | 950 | 12 h | Phase Purity | Pure MTO | |
| ZnAl₂O₄:Cr³⁺ (ZAO:Cr³⁺) [17] | Direct Solid-State | None | Not Specified | Not Specified | Photoluminescence (PL) Intensity | Baseline (1x) |
| Flux-Assisted | NH₄Cl | Not Specified | Not Specified | Photoluminescence (PL) Intensity | 3.82x enhancement | |
| Flux-Assisted | Carbon Dots (CDs) | Not Specified | Not Specified | Photoluminescence (PL) Intensity | 11.23x enhancement | |
| Flux+CDs Assisted | NH₄Cl + CDs | Not Specified | Not Specified | Photoluminescence (PL) Intensity | 17.25x enhancement |
The data reveals two primary advantages of flux-assisted synthesis. First, it enables a dramatic reduction in synthesis temperature, as seen with MTO:Mn⁴⁺ where the temperature dropped by 450°C. This lowers energy consumption and can make synthesis feasible for thermally sensitive compounds. Second, fluxes can significantly enhance functional properties, such as the over 17-fold increase in photoluminescence intensity for ZAO:Cr³⁺, which is critical for applications in LEDs, sensors, and forensic science [17] [3].
Detailed Experimental Protocols
To ensure reproducibility and provide a clear technical roadmap, this section outlines standardized protocols for both direct and flux-assisted synthesis, referencing the studies from Table 1.
Protocol 1: Direct Solid-State Synthesis
This is the conventional method for synthesizing polycrystalline materials from solid precursors.
- Key Reagents: High-purity metal oxides, carbonates, or other stable solid precursors.
- Equipment: Agate mortar and pestle or ball mill, high-temperature furnace (capable of reaching up to 1400-1500°C), alumina or platinum crucibles.
- Procedure:
- Weighing & Mixing: Weigh starting precursors according to the desired stoichiometric ratio of the target compound. For example, to synthesize Mg₂TiO₄:Mn⁴⁺, use MgO, TiO₂, and MnCO₃ [3].
- Grinding: Transfer the powder mixture to an agate mortar. Add a moderate amount of inert grinding medium (e.g., acetone or ethanol) to create a slurry and grind thoroughly for 30-60 minutes to achieve a homogeneous mixture and increase surface contact.
- Calcination: Transfer the ground mixture to a suitable crucible and place it in a furnace. Heat to an intermediate temperature (e.g., 800-1000°C) for several hours to decompose carbonates or hydroxides and initiate solid-state diffusion.
- Intermediate Grinding: After calcination, cool the sample to room temperature, regrind to eliminate aggregates, and ensure homogeneity.
- Sintering: Return the powder to the furnace for the final high-temperature reaction (e.g., 1300-1400°C for MTO:Mn⁴⁺). Maintain this temperature for a prolonged period (e.g., 12 hours) to complete the reaction and achieve crystallinity [3].
- Cooling: Allow the furnace to cool naturally to room temperature.
Protocol 2: Flux-Assisted Solid-State Synthesis
This method introduces a low-melting-point flux agent to facilitate the reaction at a lower temperature.
- Key Reagents: All reagents from Protocol 1, plus a flux agent (e.g., LiCl, NH₄Cl, NaCl, KCl).
- Equipment: Same as Protocol 1.
- Procedure:
- Weighing & Mixing: Weigh the stoichiometric precursors as in Protocol 1.
- Initial Grinding: Grind the precursors without the flux agent, often with a solvent, to achieve a homogeneous mixture.
- Flux Addition: Add a specific percentage by weight (e.g., 50 wt% LiCl for MTO:Mn⁴⁺ [3]) of the flux agent to the ground precursor mixture. Mix rapidly and homogeneously.
- Sintering: Transfer the mixture to a crucible and heat in a furnace. The sintering temperature is set above the melting point of the flux but significantly below the direct synthesis temperature (e.g., 950°C for MTO:Mn⁴⁺ with LiCl). The holding time is typically similar (e.g., 12 hours) [3]. The flux melts, creating a liquid medium that enhances mass transport.
- Cooling & Washing: After sintering and cooling, the product is often a solidified mass. Wash the product repeatedly with deionized water or a suitable solvent (e.g., hot water for LiCl) to completely dissolve and remove the residual flux salts [3].
- Drying: Dry the purified powder at a moderate temperature (e.g., 80-120°C) to obtain the final product.
The following diagram visualizes the key stages and decision points in the flux-assisted synthesis workflow:
The Scientist's Toolkit: Key Reagents and Materials
Successful execution of solid-state reactions requires specific reagents and instruments. The following table details essential items and their functions, with a focus on flux chemistry.
Table 2: Essential Research Reagent Solutions for Solid-State Synthesis
| Category | Item / Reagent | Specification / Purity | Primary Function in Synthesis |
|---|---|---|---|
| Precursors | Metal Oxides (e.g., MgO, TiO₂, Al₂O₃) | 99.9% (Nanopowders preferred) | Provide metal cations for the target crystal lattice [3]. |
| Carbonates (e.g., MnCO₃) | 99.95% | Source of metal cations; decomposes to release oxide upon heating [3]. | |
| Flux Agents | Chloride Salts (LiCl, NH₄Cl, KCl, NaCl) | Analytical Grade (AR) | Low-melting-point fluxes to create a liquid reaction medium, enhancing diffusion and reducing synthesis temperature [17] [3]. |
| Carbon Dots (CDs) | Synthesized from precursors like citric acid and urea | Novel carbon-based modifiers that can dramatically enhance luminescent properties when composited with phosphors [17]. | |
| Equipment | High-Temperature Furnace | Capable of ≥1500°C | Provides controlled high-temperature environment for reaction and sintering. |
| Agate Mortar & Pestle | -- | For manual grinding and homogenization of powder mixtures. | |
| Ball Mill | -- | For mechanical grinding and achieving uniform particle size. | |
| Alumina Crucibles | High-purity >99% | Inert containers for holding samples during high-temperature treatment. |
The strategic control of temperature, time, and atmosphere is fundamental to directing solid-state synthesis outcomes. As evidenced by comparative data, flux-assisted methods offer a powerful alternative to direct solid-state reactions, enabling dramatic reductions in synthesis temperature (e.g., from 1400°C to 950°C) and significant enhancements in material performance (e.g., >17x PL intensity). The choice between methods should be guided by a thermodynamic analysis of the reaction system. When kinetic barriers dominate, the use of a flux provides a liquid-phase pathway to overcome diffusion limitations and nucleation barriers, facilitating the formation of phase-pure, high-performance materials. Integrating this understanding of critical process parameters with the strategic application of flux chemistry provides researchers with a robust framework for accelerating the discovery and optimization of advanced inorganic materials.
Historical Context and Evolution of Solid-State Synthesis Techniques
Solid-state synthesis represents a cornerstone of materials science, providing the fundamental techniques for creating a vast array of inorganic compounds essential for modern technology. This comparative guide examines the evolution of these techniques, focusing specifically on the distinction between conventional solid-state reactions and the flux method, a specialized approach for growing high-quality crystals. Within the broader context of materials research, understanding the methodological nuances between these approaches is crucial for selecting appropriate synthesis pathways for target materials, particularly in advanced applications such as energy storage and superconductivity. This analysis provides researchers with a structured comparison of these techniques, supported by experimental data and protocols, to inform synthesis strategy decisions in both academic and industrial settings.
The foundational principle of solid-state synthesis involves high-temperature heating of solid precursor powders to facilitate atomic diffusion and subsequent chemical reaction, yielding the desired product phase [19]. This approach, often termed the ceramic method, has been widely utilized for decades due to its conceptual simplicity and directness. In contrast, the flux method, a subsidiary of solid-state reaction, introduces a liquid medium (the flux) to dissolve the reactants at elevated temperatures, enabling crystal growth from the supersaturated solution upon cooling [20]. This technique is particularly valuable for incongruent melting systems and for producing bulk single crystals.
Historical Development and Fundamental Principles
Conventional Solid-State Synthesis
The traditional solid-state route typically involves the mechanical homogenization of solid precursor compounds, followed by calcination at high temperatures, often with intermediate grinding steps to improve reactivity and phase purity [19]. A modern adaptation of this method incorporates mechanochemical activation using planetary ball mills, which applies intense mechanical forces to reduce particle size, create fresh reactive surfaces, and decrease the required synthesis temperature [19]. This approach was used in the synthesis of ZnFe2O4, where iron(III) oxide and zinc oxide were homogenized in an agate mortar and mechanically activated before thermal treatment [19].
The Flux Method
The flux method, also known as flux growth or solution growth, is designed to produce bulk single crystals from systems exhibiting incongruent melting [20]. The process involves dissolving the reactant materials in a suitable flux at a temperature exceeding its melting point, followed by slow cooling to promote crystallization from the supersaturated solution. Key advantages include the ability to grow crystals at temperatures below their melting points and the potential for achieving high crystallinity. The method has been instrumental in developing materials like Y-123 phase superconductors, where a common solvent is a "self-flux" composed of CuO and BaO [20]. Other successful fluxes include alkali metal chlorides (NaCl, KCl), In2O3, B2O3, and NaAs [20].
Table 1: Historical Milestones in Solid-State Synthesis Techniques
| Time Period | Solid-State Method Developments | Flux Method Developments |
|---|---|---|
| Early-Mid 20th Century | Establishment of basic ceramic processing; manual grinding and firing. | Early use of molten salts for crystal growth of oxides and sulfides. |
| Late 20th Century | Introduction of controlled atmosphere furnaces; use of high-purity precursors. | Critical development for high-temperature superconductors (e.g., Y-123) [20]. |
| 21st Century | Integration of mechanochemical activation [19]; automated processes; text-mining of synthesis recipes [21]. | Expansion to iron-based pnictides; exploration of novel flux agents and reductants [20]. |
Comparative Analysis: Methodologies and Experimental Protocols
Direct Solid-State Reaction Workflow
The synthesis of materials like P2–Na0.67Mn0.5Fe0.5O2 or ZnFe2O4 via the solid-state route follows a systematic protocol [22] [19]:
- Precursor Preparation: Stoichiometric amounts of precursor oxides or carbonates (e.g., Na2CO3, MnO2, Fe2O3, ZnO) are accurately weighed.
- Homogenization: Precursors are mixed thoroughly, typically using an agate mortar and pestle or a ball mill, to ensure intimate contact and compositional uniformity.
- Mechanochemical Activation (Optional but common): The mixed powder is subjected to high-energy milling in a planetary ball mill. For example, the "Aktivator-2 SL" at 1380 rpm for 30 minutes using zirconium oxide grinding media [19].
- Calcination: The homogenized powder is pressed into pellets to increase inter-particle contact and heated in a muffle furnace at high temperatures (e.g., 700–1000°C) for several hours with a controlled heating rate (e.g., 10°C/min) [19].
- Post-processing: The resulting solid is ground again into a fine powder for use as an electrode material.
Flux Method Synthesis Workflow
The synthesis of single crystals, such as NaFe1-xCoxAs or Y-123, via the flux method involves a different set of critical steps [20]:
- Flux and Precursor Preparation: Selection of an appropriate flux (e.g., NaAs, CuO/BaO self-flux, KCl-NaCl) and thorough mixing with reactant powders in a specific molar ratio (e.g., NaAs flux synthesized at 200°C in a sealed vacuum Nb tube).
- Crucible Selection: Placement of the mixture in a crucible material resistant to flux corrosion, such as yttria-stabilized zirconia (YSZ), yttria (Y2O3), or sealed tantalum tubes under an inert atmosphere [20].
- Heating and Dissolution: Heating the mixture in a furnace to a temperature above the flux's melting point to fully dissolve the reactants, forming a homogeneous solution.
- Crystal Growth: Slow cooling of the saturated solution at a controlled rate (e.g., below 0.5°C/h for Y-123) to promote the nucleation and growth of large, high-quality crystals [20].
- Flux Removal: Separation of the grown crystals from the solidified flux matrix, often achieved by decanting while hot or by washing with an appropriate solvent (e.g., water for chloride fluxes).
Performance Comparison and Experimental Data
The choice of synthesis method profoundly impacts the morphological, structural, and functional properties of the final material. A direct comparison between solid-state and spray-drying (a wet-chemical method related to the flux approach in its solution-based nature) for Mg-doped P2–Na0.67Fe0.5Mn0.5O2 cathode material highlights these differences starkly [22].
Table 2: Synthesis Method Impact on Material Properties and Electrochemical Performance [22]
| Characteristic | Solid-State Synthesis | Spray-Drying Synthesis | Implication for Application |
|---|---|---|---|
| Na+ Diffusion Coefficient (DNa+) | ~10⁻¹⁰ cm² s⁻¹ | ~10⁻⁸ cm² s⁻¹ | Ionic conductivity more than two orders of magnitude higher for spray-dried material. |
| Capacity Retention (after 100 cycles @ 1C) | 72% | 81% | Superior cycling stability for the solution-based method. |
| Morphology & Microstructure | Irregular particles, less uniform | Regular spherical morphology, optimized for cathodes | Better electrode kinetics and contact with electrolyte for spray-dried materials. |
| Mg-doping benefit on P2–Na0.67Mn0.5Fe0.5O2 | — | Capacity retention improved from 39% to 69% after 200 cycles at 1C | Effective cation substitution is enhanced in synthesis routes producing homogenous materials. |
Furthermore, in the synthesis of ZnFe2O4, the combined sol-gel and solid-state method yielded materials with different electrophysical properties, including ionic conductivity, compared to those produced by the classical ceramic technique, underscoring the importance of the synthesis pathway on the final material's performance in battery applications [19].
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful synthesis requires careful selection of starting materials, flux agents, and equipment. The following table details key components used in the methodologies discussed.
Table 3: Essential Research Reagents and Materials for Solid-State and Flux Synthesis
| Item Name | Function/Application | Specific Examples |
|---|---|---|
| Precursor Oxides/Carbonates | Source of metal cations in the final compound. | Na₂CO₃, MnO₂, Fe₂O₃, ZnO, NiO [22] [19]. |
| Flux Agents | Solvent medium for crystal growth; lowers synthesis temperature. | CuO-BaO (self-flux for Y-123), NaCl-KCl, NaAs (for iron pnictides) [20]. |
| Inert Crucibles | Contain reaction mixture; must resist corrosion by flux at high T. | Yttria-Stabilized Zirconia (YSZ), Tantalum (Ta) tubes, Gold (Au) [20]. |
| Planetary Ball Mill | Mechanochemical activation; achieves homogenization and particle size reduction. | Zirconium oxide-lined drums and balls ("Aktivator-2 SL") [19]. |
| Muffle Furnace | High-temperature calcination and crystal growth. | Capable of controlled heating rates (e.g., 10°C/min) and sustained high temperatures [19]. |
| Reductants (for some flux methods) | Lowers reaction temperature and controls morphology. | Metal elements (Sn, In, Zn, Ga) [20]. |
The evolution of solid-state synthesis techniques has provided materials scientists with a diverse toolkit, ranging from the straightforward conventional solid-state reaction to the more specialized flux method. The comparative data clearly demonstrates that the synthesis route is not merely a procedural step but a critical determinant of a material's ultimate properties and performance. The solid-state method offers simplicity and is well-suited for producing polycrystalline powders in large quantities. In contrast, the flux method is indispensable for growing high-quality single crystals, particularly of complex or incongruently melting compounds, albeit with challenges in flux removal and potential contamination.
The ongoing integration of advanced methods like mechanochemical activation and data-driven approaches, such as text-mining of synthesis recipes [21], promises to further refine these techniques. For researchers, the selection between direct solid-state and flux synthesis must be guided by the target material's crystal structure, desired morphology, and intended application, with the understanding that optimization of the synthesis parameters is often the key to unlocking high performance.
Practical Implementation: Step-by-Step Protocols for Material Synthesis
The conventional solid-state reaction route is a foundational and widely adopted method for preparing polycrystalline inorganic solids, including complex oxides, battery electrode materials, and phosphors [23] [24]. This method involves the chemical transformation of a mixture of solid starting materials, such as simple oxides, carbonates, or other metal salts, into a new solid composition through high-temperature heating [23]. The process is characterized by its reliance on solid-state diffusion of ions, which features inherently slow kinetic rates [23]. Despite its apparent simplicity, the solid-state reaction is a complex process where outcomes are influenced by numerous variables, from precursor selection to the final heat treatment [25]. Its principal advantages include relatively low cost, simple apparatus requirements, and the capability for large-volume powder production [23]. However, challenges such as powder agglomeration, limited homogeneity, and the formation of unwanted phases are recognized drawbacks [23]. This guide details the standard protocol and contrasts it with the flux method, providing a direct comparison for researchers in the field.
Detailed Experimental Protocol
Reagent Selection and Preparation
The initial step involves the careful selection and preparation of solid reactants. Common precursors include binary oxides (e.g., Co3O4, PbO, ZrO2), carbonates (e.g., Li2CO3), and other metal salts (nitrates, hydroxides, oxalates) [23] [26]. The selection is based on the desired final composition and their reactivity.
- Drying and Weighing: Precursor powders should be thoroughly dried before use. They are then weighed in the required stoichiometric amounts. To compensate for the volatilization of certain components (e.g., PbO or Li2CO3) at high temperatures, a slight excess (e.g., 1-5%) of the volatile component is often incorporated [26].
- Stoichiometry Considerations: For instance, in the synthesis of LiCoO2 (LCO), a 5% excess of Li2CO3 is used to account for lithium loss during calcination [26].
Mixing and Homogenization
After weighing, the powders must be mixed to achieve a homogeneous distribution of reactants, which is critical as reactions initiate at the points of contact between particles [23] [26].
- Manual Mixing: For small quantities (<20g), manual mixing using an agate mortar and pestle is typical. A volatile organic liquid like acetone or alcohol is often added to aid in forming a paste and improving homogenization. The liquid evaporates completely after 10-15 minutes of grinding [24].
- Mechanical Mixing: For larger quantities or to enhance reproducibility, mechanical methods like ball milling or planetary centrifugal mixing are employed [26] [24]. Counterintuitively, one study on LCO synthesis found that mechanical mixing is not always superior to physical grinding; improper mechanical mixing can sometimes lead to detrimental phase segregation, highlighting that the process must be optimized [26]. The goal is to maximize the interfacial contact area between precursor powders to facilitate the solid-state reaction [26].
Container Selection and Pelletizing
- Container Material: The reaction mixture is heated in a container that must be chemically inert under the chosen conditions. Platinum and gold are commonly used for their high-temperature stability. For lower-temperature reactions (below 600–700 °C), nickel crucibles can be suitable [24].
- Pelletizing: Before heating, the mixed powder is often pressed into pellets. This increases the area of contact between the reactant grains, improving reaction kinetics and yield [24].
Heat Treatment: Calcination
Calcination is the core step where the chemical reaction occurs. The heating program is tailored based on the reactivity of the precursors and the target material.
- Temperature and Atmosphere: Reactions typically require temperatures between 1000 °C and 1500 °C [24]. The atmosphere (air, oxygen, argon) is controlled based on material requirements. For oxide materials like LiCoO2, an oxygen atmosphere is used [26].
- Multi-step Annealing and Intermediate Milling: A single heating step often yields impure phases. The standard protocol involves several annealing steps with multiple intermediate milling stages. The initial calcination (e.g., at 750°C for 6 hours) forms the desired phase, followed by grinding, and then a final sintering at a higher temperature (e.g., 1250°C for 5 hours) [23]. Intermediate milling breaks down the product, increases powder homogeneity, decreases particle size, and makes the powder more reactive (sinter-active) for subsequent heating [23].
- Heating Profile: A two-step heating process is frequently used. For example, in LCO synthesis, the precursors are first heated to a moderate temperature (e.g., 500°C) to decompose carbonates, followed by a higher-temperature calcination (e.g., 900°C) to crystallize the target phase [26].
Product Analysis
The final product is analyzed to confirm its phase purity, morphology, and chemical composition. Standard techniques include:
- X-ray Diffraction (XRD): For phase identification and crystal structure determination [24] [27].
- Scanning Electron Microscopy (SEM): To examine particle size, morphology, and surface structure [24] [27].
- Raman Spectroscopy: Provides complementary information on molecular vibrations and can confirm phase formation and detect impurities [27].
- Energy Dispersive Spectroscopy (EDS): Used for semi-quantitative elemental analysis [28].
The following workflow diagram summarizes the key stages of the conventional solid-state reaction protocol.
The Scientist's Toolkit: Essential Research Reagents
Table 1: Key reagents and equipment used in conventional solid-state reactions.
| Item Name | Function/Description | Common Examples |
|---|---|---|
| Oxide Precursors | Source of metal cations in the reaction. | Co₃O₄, PbO, ZrO₂ [26] |
| Carbonate Precursors | Source of alkali/alkaline earth metals and Li. | Li₂CO₃ [26] |
| Mortar and Pestle | For manual mixing and grinding of precursor powders. | Agate tool [24] |
| Planetary Centrifugal Mixer/Ball Mill | For mechanical mixing/milling to enhance homogeneity and reduce particle size. | AR-100 (THINKY Corp.) [26] |
| Calcination Crucible | High-temperature container for the reaction. | Platinum, gold, or alumina boats [24] |
| Tube Furnace | Programmable furnace for controlled heating under specific atmospheres. | Nabertherm furnace [27] |
Performance Comparison: Conventional vs. Flux Synthesis
The conventional solid-state method is often compared to alternative synthesis routes. The flux method, which uses a molten reactant (the flux) in substantial excess as a solvent, provides a distinct approach with different outcomes [28].
Table 2: Comparison between conventional solid-state and flux reaction methods.
| Parameter | Conventional Solid-State Reaction | Flux Synthesis Method |
|---|---|---|
| Basic Principle | Direct reaction between solid precursors via diffusion [23]. | Reaction in a molten solvent (flux) that dissolves reactants [28]. |
| Typical Temperature | High (1000–1500°C) [24]. | Lower (e.g., 1000°C), limited by flux melting point [28]. |
| Reaction Kinetics | Slow, diffusion-controlled [23]. | Enhanced due to dissolution in liquid medium [28]. |
| Product Morphology | Agglomerated powders, irregular particles [23]. | Often larger, well-formed crystals [28]. |
| Homogeneity | Limited; requires multiple grinding steps [23]. | Can be higher due to mixing in a liquid state [28]. |
| Key Advantages | Simple, inexpensive, suitable for bulk production [23]. | Can yield high-quality crystals, access metastable phases [28]. |
| Key Disadvantages | High agglomeration, limited homogeneity, may require high temperatures [23]. | Flux incorporation into product possible, requires separation [28]. |
The flux method's use of a reactive solvent, such as molten aluminum, can facilitate the formation of single crystals and can sometimes yield phases not easily accessible through the conventional route [28]. For instance, in the synthesis of rare-earth aluminum germanides (REAl₃₋ₓGeₓ), the flux environment allowed for the formation of phases with specific disordered structures [28].
Experimental Data from Cited Studies
Case Study: Synthesis of LiCoO₂ (LCO) Cathode Material
A comparative study investigated the effect of precursor mixing on the properties of LCO.
- Methodology: Li₂CO₃ and Co₃O₄ precursors were mixed via two processes: (1) physical mixing (PM) with a mortar and pestle, and (2) mechanical mixing (MM) with a planetary centrifugal mixer. The mixed powders were then calcined using a two-step heating process (500°C followed by 900°C) [26].
- Results and Data:
- Morphology: The PM precursor showed irregular particles, while the MM precursor exhibited a more regular distribution of finer particles [26].
- Electrochemical Performance: Despite finer mixing, the MM-LCO electrode showed poorer performance with a specific capacity of ~135 mAh/g after 50 cycles, compared to ~150 mAh/g for PM-LCO. This was attributed to lithium loss in the MM precursor due to a more intimate contact with Co₃O₄, which catalyzed the premature decomposition of Li₂CO₃ [26].
- Conclusion: This study demonstrates that a routine process like mechanical mixing is not universally beneficial and must be optimized, as non-optimal conditions can be detrimental to the final material's performance [26].
Case Study: Synthesis of Cu₂FeSnS₄ (CFTS) Powders
Research on CFTS highlights the profound impact of calcination on material properties.
- Methodology: CFTS was synthesized via a solid-state reaction from elemental Cu, Fe, Sn, and S. The resulting powder was then calcined at different temperatures (800°C, 900°C, and 1000°C) [27].
- Results and Data:
- Crystallinity: XRD and Raman spectroscopy confirmed that the crystalline quality of the CFTS powders improved with increasing calcination temperature [27].
- Optical Properties: The optical band gap of the powder was found to decrease with higher calcination temperatures, from 1.36 eV (uncalcined) to 1.30 eV (calcined at 1000°C), tailoring the material for specific optoelectronic applications [27].
- Conclusion: Post-synthesis calcination is an effective method for enhancing the structural and optical properties of materials prepared by solid-state reaction [27].
The logical relationship between synthesis parameters, intermediate phases, and the final product's performance is summarized below.
In solid-state synthesis, a flux is a solvent medium that facilitates the reaction between solid reactants at elevated temperatures. The flux method is a powerful alternative to direct solid-state reaction, often yielding products with improved crystallinity, phase purity, and controlled morphology at lower temperatures. This guide provides a comparative analysis of three chloride-based flux systems: CsCl, CaCl₂, and LiCl/MgCl₂ mixtures, offering researchers a framework for selecting the appropriate agent based on specific synthesis goals.
Comparative Analysis of Flux Agents
The table below summarizes the key performance characteristics and experimental parameters for CaCl₂, LiCl, and MgCl₂ as evidenced by recent research. It is important to note that while the search results provide robust data for CaCl₂, LiCl, and MgCl₂, no specific experimental data for CsCl was identified in the consulted sources. Researchers are advised to consult specialized databases or prior literature for its properties.
Table 1: Comparative Performance of Chloride-Based Flux Agents in Solid-State Synthesis
| Flux Agent | Optimal Processing Temperature & Conditions | Key Effects on Product & Performance | Quantitative Efficacy Data |
|---|---|---|---|
| LiCl | ~950°C for 12 hours (50 wt% flux) [3] | Lowers synthesis temperature dramatically; enables pure phase formation; improves particle morphology and photoluminescence intensity [3]. | Pure Mg₂TiO₄:Mn⁴⁺ formed at 950°C with LiCl, versus persistent impurities at 1400°C without flux [3]. |
| CaCl₂ | Serves as a component in a LiCl-CaCl₂ mixed flux [30] | Promotes crystal growth in complex systems; enables synthesis of high-entropy materials [30]. | Facilitated growth of high-entropy oxysulfide Gd₂/₃Y₁/₃Sm₁/₃Tb₁/₃Ho₁/₃Ti₂O₅S₂ single crystals [30]. |
| MgCl₂ | Not specified for solid-state synthesis in available data. | Reference data primarily from environmental/industrial applications suggests potential corrosivity [31]. | In dust control, ~40-75% more product is required vs. CaCl₂ for equivalent performance [31]. NACE data indicates MgCl₂ can be 10x more corrosive to mild steel than CaCl₂ [31]. |
Detailed Experimental Protocols
Protocol 1: Solid-State Synthesis Using LiCl Flux for Phosphors
This protocol, adapted from the synthesis of Mg₂TiO₄:Mn⁴⁺ (MTO:Mn⁴⁺) phosphor, details the use of LiCl flux to lower reaction temperature and eliminate impurity phases [3].
- Materials:
- Precursors: Anatase TiO₂ (99.8%), MgO (99.9%), MnCO₃ (99.95%).
- Flux: Anhydrous LiCl.
- Solvent: Anhydrous ethanol.
- Methodology:
- Weighing and Grinding: Weigh TiO₂, MgO, and MnCO₃ in stoichiometric proportions for the target compound (e.g., MTO:0.2% Mn⁴⁺). Place the powder mixture in an agate mortar, add anhydrous ethanol, and grind thoroughly until homogenous.
- Flux Addition: Add the LiCl flux (e.g., 50% by weight of the precursor mass) to the mixture and grind again rapidly to ensure homogeneous mixing.
- Sintering: Transfer the final mixture to a corundum crucible. Place the crucible in a muffle furnace and sinter at the target temperature (e.g., 800–1050°C) for a hold time of 12 hours.
- Post-Synthesis Processing: After the furnace has cooled naturally to room temperature, remove the product. Wash the sintered product several times with deionized water to completely dissolve and remove residual LiCl salts. Finally, dry the purified product.
- Key Observations: Without LiCl flux, the Mg₂TiO₄ phase was not pure even at 1400°C, with significant MgTiO₃ impurities present. The addition of 50 wt% LiCl enabled the formation of a pure phase at 950°C. Excessive flux can lead to secondary phases.
Protocol 2: Flux Growth from a LiCl-CaCl₂ Mixture for Oxysulfides
This protocol is derived from the synthesis of high-entropy layered perovskite-type oxysulfide single crystals, demonstrating the use of a mixed chloride flux for growing complex crystals [30].
- Materials:
- Precursors: Rare-earth oxides or other relevant metal compounds.
- Flux: A mixture of LiCl and CaCl₂.
- Methodology:
- Precursor and Flux Preparation: Combine the solid reactant precursors with the mixed LiCl-CaCl₂ flux in a suitable crucible.
- Heating and Crystal Growth: Heat the mixture to a high temperature where the flux melts, creating a liquid solution supersaturated with the reactants. Maintain this temperature to allow for the slow growth of single crystals from the flux melt.
- Cooling and Separation: Slowly cool the melt to promote further crystal growth. Once cooled, separate the crystals from the solidified flux matrix, often by dissolving the water-soluble chloride flux in deionized water.
- Key Observations: The LiCl-CaCl₂ flux permitted the formation of well-defined, platy single crystals (~1 µm in size) where the multiple rare-earth elements were uniformly distributed throughout the crystal structure, a critical requirement for high-entropy materials [30].
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents and Their Functions in Flux-Assisted Solid-State Synthesis
| Reagent / Material | Primary Function in Synthesis |
|---|---|
| Chloride Salts (LiCl, CaCl₂, etc.) | Acts as a flux; dissolves solid reactants at high temperature to enhance ion diffusion and reaction kinetics, lowering synthesis temperature and improving crystallinity [30] [3]. |
| Agate Mortar and Pestle | Used for the mechanical grinding and mixing of solid precursors and flux to achieve a homogeneous starting mixture, which is critical for a uniform reaction [3]. |
| Corundum (Al₂O₃) Crucible | A high-temperature vessel that is chemically inert and can withstand sintering temperatures often exceeding 1000°C without reacting with the sample or flux [3]. |
| Deionized Water | Used for the post-synthesis washing step to dissolve and remove the water-soluble chloride flux from the final product after sintering [3]. |
Decision Workflow for Flux Selection
The following diagram illustrates a logical pathway for selecting an appropriate flux based on synthesis objectives, derived from the experimental findings.
The selection of an appropriate flux is a critical determinant in the success of a solid-state synthesis. While direct solid-state reactions are simple, the use of fluxes like LiCl and CaCl₂ can overcome significant kinetic barriers.
- LiCl has been demonstrated as a highly effective flux for drastically reducing the synthesis temperature and achieving phase-pure products in oxide systems.
- CaCl₂, particularly in mixtures, facilitates the crystal growth of complex, high-entropy materials.
- MgCl₂, based on comparative data, may present challenges related to corrosivity, a factor that must be considered for reactor compatibility.
- CsCl remains a flux of interest, though current data was unavailable in this analysis.
Researchers are encouraged to use this guide as a starting point, supplementing it with further experimental data and literature reviews to tailor the flux selection to their specific chemical system.
The pursuit of efficient and stable photocatalysts for solar-driven water splitting is a central goal in the quest for renewable hydrogen production. Among the promising materials, the oxysulfide Y₂Ti₂O₅S₂ (YTOS) has garnered significant attention due to its narrow bandgap (enabling light absorption up to ~650 nm) and outstanding thermal and chemical stability compared to pure sulfides [32] [33]. These properties make it a potential candidate for high-performance photocatalytic applications. However, the practical application of YTOS is heavily constrained by the limitations of its conventional synthesis methods, which often involve sealed quartz tubes, long reaction times, and challenges in scaling up [32] [33].
This case study objectively analyzes a novel synthesis approach for YTOS using carbon disulfide (CS₂) sulfurization, framing it within a broader thesis comparing direct solid-state reaction and flux-assisted methods. We present a direct performance comparison with established alternatives, supported by experimental data and detailed protocols, to provide researchers with a clear understanding of this emerging synthetic pathway.
Established YTOS Synthesis Methods and Their Limitations
To appreciate the advancements offered by CS₂ sulfurization, it is essential to first understand the drawbacks of existing synthesis routes.
Solid-State Reaction (SSR): The conventional SSR method involves mixing precursor oxides and sulfides (e.g., Y₂O₃, Y₂S₃, and TiO₂) in a sealed quartz tube under vacuum [33]. This process requires extended calcination times, typically longer than four days, to complete the reaction due to low mass transfer rates between solid reactants [33]. The resulting YTOS crystals are often characterized by large particle sizes (micrometers to tens of micrometers) and a high density of bulk defects that act as recombination centers for photogenerated charge carriers, thereby limiting photocatalytic performance [33] [34].
H₂S Sulfurization: This method offers an alternative by using gaseous H₂S to convert oxide precursors into YTOS. A significant advantage is the substantial reduction in reaction time (to about 1 hour) and the avoidance of sealed-tube reactions, making it more amenable to scale-up [33]. However, it necessitates a high reaction temperature (around 1150 °C) and often yields materials with reduced crystallinity and a high defect density, partly due to hydrogen byproduct formation during sulfidation. These factors typically result in inferior photocatalytic activity compared to SSR-synthesized YTOS [32] [33].
Flux-Assisted Synthesis: The introduction of a molten salt flux (e.g., CaCl₂, CsCl) to either the SSR or sulfurization methods enhances mass transfer by dissolving solid reactants and facilitating rapid ion migration [33] [34]. This approach leads to smaller particle sizes, enhanced crystallinity, and reduced defect densities, which collectively contribute to improved photocatalytic performance for both hydrogen and oxygen evolution reactions [34]. Nevertheless, when used within a sealed-tube SSR process, it still faces scalability challenges.
CS₂ Sulfurization: A Novel Synthetic Approach
The CS₂ sulfurization method represents a hybrid approach that combines the use of a new sulfurization reagent with the benefits of a flux to address the limitations of previous techniques [32] [33].
This synthesis employs carbon disulfide (CS₂) as the sulfurizing agent in the presence of a CaCl₂ flux [33]. The high reactivity of CS₂ allows the formation of YTOS from oxide precursors at a significantly lower temperature (800 °C) than H₂S sulfurization [32]. The concurrent use of a flux enhances mass transfer, ensuring high crystallinity and preventing the formation of intermediate phases or binary sulfides [33].
Detailed Experimental Protocol
The following workflow and detailed protocol are based on the experimental section of the primary research article [33].
Key Materials and Reagents:
- Precursors: Y₂O₃ (99%), TiO₂ (rutile phase, 99.99%). Alternatively, a mixture of Y₂O₃, Y₂S₃, and TiO₂, or pre-synthesized Y₂Ti₂O₇ can be used [33].
- Flux: Anhydrous CaCl₂ (500 wt% relative to the precursor mass) [33].
- Sulfurizing Agent: Liquid CS₂ (99% purity) [33].
- Inert Atmosphere: High-purity N₂ gas.
- Equipment: Horizontal tube furnace, alumina boat, agate mortar, glovebox.
Step-by-Step Procedure:
- Precursor Mixing: In a N₂-filled glovebox, the chosen precursors are thoroughly mixed with the CaCl₂ flux using an agate mortar for 30 minutes [33].
- Reactor Setup: Approximately 3.0 g of the mixture is transferred to an alumina boat, which is then placed in a horizontal tube furnace. A stream of N₂ gas is bubbled through the liquid CS₂ (maintained at 10°C for stable evaporation) to create a CS₂/N₂ gas mixture, which is fed into the reactor [33].
- Thermal Treatment: The furnace is heated at 10°C min⁻¹ to 500°C, then at 5°C min⁻¹ to the target temperature of 800°C, and held there for 3 hours [33].
- Cooling and Purge: After the reaction, the sample is cooled naturally to 300°C. The CS₂ flow is stopped, and a pure N₂ flow is maintained for an additional 4 hours to remove any residual CS₂ [33].
- Product Recovery: The resulting solid is sonicated in distilled water to dissolve and remove the water-soluble CaCl₂ flux. The final YTOS powder is collected by filtration and dried [33].
Performance Comparison and Discussion
The photocatalytic performance of YTOS synthesized via CS₂ sulfurization has been quantitatively evaluated, primarily through sacrificial hydrogen evolution reactions.
Comparative Photocatalytic Activity
The following table summarizes the key advantages of the CS₂ sulfurization method against established synthesis techniques, based on reported experimental data [32] [33].
Table 1: Comparative Analysis of YTOS Synthesis Methods
| Synthesis Method | Reaction Temperature | Reaction Time | Key Characteristics | Reported Photocatalytic H₂ Evolution Activity |
|---|---|---|---|---|
| Solid-State Reaction (SSR) | ~1000°C [33] | >4 days [33] | Large particles, high defect density [33] | Baseline for comparison |
| H₂S Sulfurization | ~1150°C [33] | ~1 hour [33] | High defect density, impurities [32] | Lower than SSR [32] |
| Flux-Assisted SSR | High (sealed tube) | Shorter than SSR [34] | Reduced particle size, enhanced crystallinity [34] | High [34] |
| CS₂ Sulfurization (with flux) | 800°C [32] | 3 hours [33] | Improved crystallinity, fewer defects [32] | Greater than SSR and H₂S methods; comparable to flux-assisted SSR [32] |
Discussion of Comparative Data
The experimental data indicates that YTOS produced via CS₂ sulfurization exhibits superior photocatalytic H₂ evolution activity compared to both conventional SSR and H₂S-synthesized YTOS [32]. Its performance is comparable to the high-quality YTOS crystals prepared by the flux-assisted method [32]. This enhanced activity is attributed to several synergistic factors:
- Reduced Synthesis Temperature: The high reactivity of CS₂ enables complete sulfurization at 800°C, which is 350°C lower than the H₂S method. This lower temperature mitigates thermal degradation and reduces the formation of defects [32] [33].
- Enhanced Crystallinity and Purity: The combination of an active gas-phase sulfur source and the mass-transfer-promoting flux facilitates the growth of well-crystallized YTOS with fewer bulk defects that would otherwise promote charge carrier recombination [32].
- Scalability Potential: Unlike sealed-tube methods, the CS₂ sulfurization process operates in an open gas-flow system, which is inherently more adaptable to continuous or large-batch production for practical applications [32].
The Scientist's Toolkit: Essential Research Reagents
The table below lists key reagents and their critical functions in the CS₂ sulfurization synthesis of YTOS, serving as a quick reference for experimental design.
Table 2: Key Research Reagent Solutions for CS₂ Sulfurization
| Reagent | Function in the Synthesis | Key Consideration |
|---|---|---|
| Carbon Disulfide (CS₂) | High-activity sulfurizing reagent; enables lower reaction temperature. | Highly flammable and toxic; requires careful handling and controlled evaporation. [32] [33] |
| Calcium Chloride (CaCl₂) | Molten salt flux; enhances mass transfer between solid reactants, improves crystallinity, and reduces particle size. | Hydroscopic; must be handled in an inert atmosphere. Water-soluble, allowing easy removal post-synthesis. [33] |
| Yttrium Oxide (Y₂O₃) & Titanium Oxide (TiO₂) | Oxide precursors for the formation of YTOS. | Purity (≥99%) and phase (e.g., rutile TiO₂) are critical for achieving phase-pure YTOS. [33] |
| Nitrogen (N₂) Gas | Creates an inert atmosphere and acts as a carrier gas for CS₂ vapor. | High purity is essential to prevent oxidation of precursors and the product during synthesis. [33] |
The synthesis of Y₂Ti₂O₅S₂ via CS₂ sulfurization represents a significant step forward in the development of practical oxysulfide photocatalysts. This method successfully addresses key limitations of traditional techniques by lowering the required reaction temperature, reducing process time, and yielding a material with high crystallinity and competitive photocatalytic performance.
Framed within the broader comparison of synthesis methods, CS₂ sulfurization emerges as a promising hybrid pathway that combines the scalability advantages of gas-phase sulfurization with the material quality benefits of flux-assisted synthesis. For researchers and scientists in the field, this approach offers a viable and efficient route to producing high-quality YTOS, thereby accelerating the optimization and application of this promising material in sustainable hydrogen production. Future work will likely focus on further optimizing the flux composition and CS₂ delivery parameters to push the performance even closer to the theoretical limits of YTOS.
The quest for advanced battery technologies beyond conventional lithium-ion systems has intensified the search for novel electrode materials. Among them, zinc ferrite (ZnFe₂O₄) spinel has emerged as a promising candidate due to its high theoretical capacity, environmental friendliness, and cost-effectiveness [19] [35]. The performance of ZnFe₂O₄ in battery applications is profoundly influenced by its synthesis route, which governs critical characteristics such as particle size, crystallinity, cation distribution, and ultimately, electrochemical behavior [19] [36]. This case study provides a comprehensive comparison of predominant synthesis methodologies—solid-state, sol-gel, and hydrothermal routes—framed within the broader context of direct solid-state reaction flux methods research. By examining detailed experimental protocols, structural properties, and electrochemical performance metrics, this guide aims to equip researchers with the necessary knowledge to select and optimize synthesis strategies for ZnFe₂O₄ in energy storage applications.
Synthesis Methodologies: A Comparative Analysis
Solid-State Synthesis
The solid-state method represents a classical ceramic processing route characterized by high-temperature treatment of precursor oxides.
Experimental Protocol: In a representative procedure, iron(III) oxide (Fe₂O₃) and zinc oxide (ZnO) powders are mixed in a 1:1 molar ratio and subjected to preliminary homogenization using an agate mortar. The mixture undergoes mechanochemical activation in a planetary ball mill equipped with zirconium oxide grinding media at 1380 rpm for 30 minutes. The activated precursor is then thermally treated in a muffle furnace with a controlled heating rate of 10°C/min. The synthesis temperature is typically determined by thermal analysis and can vary across a wide range (e.g., 750–1100°C) to optimize phase formation [19] [37].
Key Characteristics: This method typically yields microscale particles with high crystallinity and well-defined spinel structure. However, it often results in larger particle sizes, broader size distributions, and lower specific surface area compared to wet-chemical methods, which can limit electrochemical performance by reducing active sites and lengthening ion diffusion paths [19].
Sol-Gel Synthesis
The sol-gel method is a versatile wet-chemical technique known for producing materials with high homogeneity and controlled stoichiometry at relatively lower temperatures.
Experimental Protocol: A combined sol-gel and solid-state finishing process has been demonstrated for ZnFe₂O₄ synthesis. Initially, chlorides of iron(III) and zinc(II) are mixed in the required molar ratio under intensive stirring. A sodium hydroxide (NaOH) solution is then introduced at room temperature to precipitate a solid precursor phase, carefully controlling the pH to 10.5 to prevent dissolution of zinc hydroxide. The resulting suspension undergoes hydrodynamic processing for 30–60 minutes, followed by vacuum filtration and washing with deionized water to remove residual salts. The filtered precursor is dried and subsequently treated in a muffle furnace under various temperature regimes to form the final crystalline spinel phase [19].
Key Characteristics: Sol-gel derived powders generally exhibit finer particle size, narrower size distribution, and higher specific surface area than solid-state synthesized materials. These morphological characteristics are advantageous for battery applications, as they provide more active sites for electrochemical reactions and shorter pathways for ion diffusion [19].
Hydrothermal Synthesis
The hydrothermal method involves crystallizing materials from aqueous solutions at elevated temperatures and pressures, offering exceptional control over particle size and morphology.
Experimental Protocol: In a standard hydrothermal synthesis, 2 mmol of zinc acetate dihydrate and 4 mmol of iron chloride hexahydrate are dissolved in 30 mL of ethylene glycol under continuous magnetic stirring. Sodium acetate is added as a fuel and mineralizer to control particle size and nucleation. The homogeneous solution is transferred to a Teflon-lined stainless steel autoclave and heated at 180°C for 24 hours. The resulting precipitate is collected via centrifugation, repeatedly washed with distilled water, and finally dried in air at 60°C to obtain the ZnFe₂O₄ powder. Variations in sodium acetate concentration (e.g., 11.5 to 62.2 mmol) enable precise tuning of nanoparticle size [35].
Key Characteristics: Hydrothermal synthesis typically produces nanoparticles with highly uniform morphology, often spherical, and exceptionally high surface area. The method allows for precise control over crystallite size through modulation of fuel concentration and reaction parameters, making it ideal for optimizing surface-dependent properties critical for electrochemical performance [35].
Table 1: Comparative Analysis of ZnFe₂O₄ Synthesis Methods
| Synthesis Parameter | Solid-State Method | Sol-Gel Method | Hydrothermal Method |
|---|---|---|---|
| Starting Materials | Metal oxides (ZnO, Fe₂O₃) [19] | Metal chlorides/nitrates, NaOH [19] | Metal acetates/chlorides, sodium acetate [35] |
| Processing Temperature | High (750–1100°C) [19] [37] | Medium (Precursor formation at RT, annealing <1000°C) [19] | Low (Reaction at 180°C) [35] |
| Reaction Time | Hours (including milling & calcination) [19] | Hours to days (including gelation & annealing) [19] | ~24 hours [35] |
| Particle Size | Micrometer scale [19] | Nanometer to sub-micrometer scale [19] | Nanoscale (tunable down to ~10 nm) [35] |
| Surface Area | Lower | Higher | Highest (increases with decreasing particle size) [35] |
| Crystallinity | High | High | High |
| Morphology Control | Limited | Moderate | Excellent (spheres, rods, etc.) [35] |
| Cost & Complexity | Low equipment cost, high energy cost | Moderate | Moderate to high |
Structural and Property Characterization
The structural and physical properties of ZnFe₂O₄ are critically dependent on the synthesis method, which influences cation distribution between tetrahedral (A) and octahedral [B] sites in the spinel lattice.
Crystal Structure and Cation Distribution: Bulk ZnFe₂O₄ is a normal spinel with Zn²⁺ occupying tetrahedral sites and Fe³⁺ occupying octahedral sites, resulting in antiferromagnetic behavior [35]. However, at the nanoscale, a mixed spinel structure often forms due to cation redistribution, where some Fe³⁺ cations migrate to tetrahedral sites and some Zn²⁺ to octahedral sites [38]. This redistribution is more pronounced in wet-chemical methods like hydrothermal and sol-gel synthesis due to lower processing temperatures and higher surface energy effects, leading to significantly enhanced magnetization compared to bulk materials [38] [37]. For instance, hydrothermally synthesized ZnFe₂O₄ can exhibit magnetization values as high as 13.4 emu/g at 20 kOe, displaying superparamagnetic behavior [39].
Particle Size and Surface Area: Wet-chemical methods consistently produce smaller particles with higher surface areas. Research shows that decreasing the size of ZnFe₂O₄ nanoparticles via modified hydrothermal conditions directly increases the Brunauer-Emmett-Teller (BET) surface area, providing more active sites for electrochemical reactions [35]. Sol-gel derived materials also demonstrate favorable electrophysical properties, including enhanced ionic conductivity, which is crucial for battery electrode performance [19].
Table 2: Structural and Magnetic Properties of ZnFe₂O₄ from Different Synthesis Routes
| Characteristic | Solid-State Derived ZnFe₂O₄ | Sol-Gel Derived ZnFe₂O₄ | Hydrothermally Derived ZnFe₂O₄ |
|---|---|---|---|
| Crystal Structure | Cubic spinel (Fd-3m) [19] | Cubic spinel (Fd-3m) [19] | Cubic spinel (Fd-3m) [35] |
| Lattice Parameter | ~8.44 Å [37] | Refined based on annealing temperature [19] | ~8.35 Å [35] |
| Cation Distribution | Predominantly normal spinel [37] | Mixed spinel (varies with temperature) [19] | Mixed spinel (inversion common) [35] [38] |
| Average Crystallite Size | Larger crystallites (>50 nm) [19] | Tunable nanoscale dimensions [19] | 10–36 nm (adjustable with fuel) [35] [38] |
| Magnetic Behavior | Weak magnetic moment (near antiferromagnetic) [37] | Ferrimagnetic/Superparamagnetic [19] | Superparamagnetic at room temperature [39] [38] |
| Magnetization (at 20 kOe) | Low (e.g., ~2 emu/g) [37] | Medium | High (e.g., 13.4 emu/g) [39] |
Performance in Battery Applications
ZnFe₂O₄ possesses a high theoretical capacity of approximately 1000 mAh g⁻¹, making it an attractive anode material for both lithium-ion and emerging zinc-ion batteries [35] [36]. Its performance is heavily influenced by the synthesis-driven structural and morphological factors.
The nanoparticle morphology achieved through sol-gel and hydrothermal routes enhances electrochemical performance through several mechanisms: (1) reduced diffusion lengths for Li⁺ or Zn²⁺ ions, improving rate capability; (2) higher surface area providing more active reaction sites; and (3) better accommodation of volume changes during charge-discharge cycling, mitigating mechanical degradation [36]. Impedance spectroscopy studies confirm that the electrophysical properties, particularly electrical ionic conductivity, differ significantly based on synthesis conditions and methods, with firing temperature being a critical parameter [19].
Furthermore, the spinel structure of ZnFe₂O₄ features a 3D network of channels that facilitate Zn²⁺ migration, providing a fundamental rationale for its promising ionic conductivity when used in metal-ion batteries [19]. Hybrid architectures, such as combining ZnFe₂O₄ with carbonaceous materials (graphene, carbon nanotubes), have shown to further enhance electrical conductivity and structural stability, leading to superior cycle life and capacity retention [36].
The Scientist's Toolkit: Essential Research Reagents
The synthesis of ZnFe₂O₄ requires specific precursors and reagents tailored to the chosen methodology.
Table 3: Key Research Reagent Solutions for ZnFe₂O₄ Synthesis
| Reagent | Typical Function | Example Application |
|---|---|---|
| Zinc Oxide (ZnO) | Solid-state precursor providing Zn²⁺ ions [19] | Solid-state reaction with Fe₂O₃ [19] |
| Iron(III) Oxide (Fe₂O₃) | Solid-state precursor providing Fe³⁺ ions [19] | Solid-state reaction with ZnO [19] |
| Zinc Nitrate/Chloride/Acetate | Soluble precursor providing Zn²⁺ ions in solution [19] [35] [39] | Co-precipitation, sol-gel, and hydrothermal synthesis |
| Iron(III) Nitrate/Chloride | Soluble precursor providing Fe³⁺ ions in solution [19] [35] [39] | Co-precipitation, sol-gel, and hydrothermal synthesis |
| Sodium Hydroxide (NaOH) | Precipitating agent to form metal hydroxides from solutions [19] | pH control and precipitation in sol-gel/co-precipitation [19] |
| Sodium Acetate | Fuel and structure-directing agent [35] | Controlling particle size and nucleation in hydrothermal synthesis [35] |
| Ethylene Glycol | Solvent and reducing agent [35] [38] | Medium for hydrothermal and solvothermal synthesis |
Synthesis Workflow Visualization
The following diagram illustrates the key decision points and procedural steps involved in selecting and executing a synthesis method for ZnFe₂O₄, from precursor selection to final application.
Synthesis Workflow for ZnFe₂O₄
The selection of a synthesis method for ZnFe₂O₄ spinel ferrites represents a critical strategic decision that directly dictates the material's structural, magnetic, and electrochemical properties. Solid-state synthesis offers simplicity and high crystallinity but yields larger particles with limited surface area. Sol-gel and co-precipitation routes provide an effective balance, producing homogeneous nanomaterials with enhanced properties suitable for battery applications. The hydrothermal method excels in delivering ultra-fine, uniform nanoparticles with the highest surface area, enabling superior electrochemical performance, albeit with more complex processing. The optimal choice depends on the specific application requirements, balancing factors such as particle size, crystallinity, processing cost, and scalability. Future research will likely focus on hybrid synthesis approaches and advanced heterostructures to further unlock the potential of ZnFe₂O₄ in next-generation energy storage systems.
Overcoming Scalability Challenges in Sealed vs. Open Reactor Systems
In materials science and chemical engineering, the choice between sealed (closed) and open reactor systems is a fundamental decision that profoundly influences the scalability, efficiency, and economic viability of processes ranging from thermal energy storage to advanced materials synthesis. Sealed reactors operate as completely isolated systems where no mass exchange with the external environment occurs, though energy transfer is permitted. These systems maintain a pure, controlled atmosphere essential for processes sensitive to contamination or requiring specific gas compositions. Conversely, open reactors allow continuous mass and energy transfer between the system and its surroundings, often utilizing ambient air or other externally supplied gases as reactive components or heat transfer media [40]. This configuration simplifies material handling but introduces potential challenges related to impurity control and environmental dependencies.
The scalability of these systems presents distinct technical challenges that become more pronounced at pilot and industrial scales. As reactor volume increases, physical phenomena such as heat transfer, mass transport, and reaction kinetics do not scale linearly, creating engineering hurdles that must be systematically addressed. For thermal energy storage applications, scalability constraints directly impact energy density, charge/discharge rates, and ultimately the economic feasibility of deploying these technologies for seasonal energy storage or industrial processes. Similarly, in materials synthesis, scalability determines the transition from laboratory-scale curiosity to commercially viable production. This guide objectively compares the performance characteristics of sealed versus open reactor configurations, with particular emphasis on quantifying how these systems address scalability challenges across different applications and scale-up stages.
Comparative Performance Analysis
Quantitative Performance Metrics
The performance divergence between sealed and open reactor systems becomes evident when examining key operational parameters across different applications. Thermochemical energy storage represents one application where both configurations have been extensively studied, revealing distinct advantages and limitations for each approach.
Table 1: Performance Comparison of Sealed vs. Open Thermochemical Reactor Systems for Thermal Energy Storage
| Performance Parameter | Sealed/Closed Reactor | Open Reactor | Measurement Context |
|---|---|---|---|
| Energy Storage Density | High (theoretical) | 1 GJ/m³ (demonstrated) | Based on MgCl₂·6H₂O; long-term storage [41] |
| Heating Power Delivery | Potentially higher | 50 W at 60°C (lab-scale) | Lab prototype with 17L MgCl₂·6H₂O [41] |
| Thermal Efficiency | Limited by heat transfer | Instantaneous COP of 12 | Coefficient of Performance during hydration [41] |
| Reaction Limitation | Gas transfer through bed | Heat transfer | Primary scaling limitation identified [40] |
| System Complexity | High (heat exchangers required) | Lower (direct air contact) | Infrastructure requirements [40] |
| Operating Temperature | Higher potential | 60°C demonstrated | Heating capacity for residential applications [41] |
The data reveals a fundamental trade-off: sealed systems offer theoretical performance advantages in energy density and operating potential, while open systems demonstrate practical implementation benefits with simpler infrastructure and proven, though modest, performance metrics at experimental scales.
Scalability Challenges by System Type
The transition from laboratory to industrial scale introduces distinct challenges for each reactor configuration. In sealed thermochemical systems, the primary limitation is gas transfer through the reactive bed, which becomes increasingly problematic as reactor dimensions expand. The permeability of the packed bed and pressure differentials available for driving gas flow do not scale favorably with increasing reactor volume, creating mass transport limitations that reduce overall reaction rates in larger systems [40]. Additionally, the requirement for integrated heat exchangers in sealed systems introduces complex engineering challenges at scale, as the surface-to-volume ratio decreases with larger reactors, potentially compromising heat transfer efficiency.
For open thermochemical systems, the dominant scaling limitation shifts to heat transfer dynamics rather than mass transport. As reactor volume increases, maintaining efficient thermal management throughout the reactive bed becomes challenging, particularly during the exothermic hydration process where localized heating can create reaction fronts that proceed unevenly through the material [40]. The use of moist air as both reactant and heat transfer fluid simplifies system architecture but introduces dependency on ambient conditions that may vary seasonally or geographically, creating operational inconsistencies at commercial scale. Furthermore, the potential for contamination or clogging from airborne particulates increases with system size and operational lifetime, requiring additional filtration or maintenance protocols [41].
Experimental Protocols and Methodologies
Thermochemical Storage Reactor Testing
The experimental protocols for evaluating thermochemical reactor performance follow standardized methodologies to ensure comparable results across different system configurations and scales. For open reactor systems, the typical experimental setup consists of a packed bed reactor containing the thermochemical material (such as MgCl₂·6H₂O), an air circulation system with controlled temperature and humidity, and comprehensive monitoring instrumentation [41].
The dehydration (charging) protocol involves circulating heated air (typically 130°C) through the reactive bed while monitoring temperature profiles across multiple points in the bed and measuring outflow humidity. The process continues until the outlet humidity approaches inlet levels, indicating complete dehydration. The hydration (discharge) protocol reverses this process, exposing the dehydrated material to moist air at ambient temperature while measuring temperature increases throughout the bed and quantifying heat output. During testing, key parameters monitored include: temperature profiles at multiple bed locations, airflow rates and humidity levels, pressure drop across the bed, and reaction front progression速度 [41].
For sealed reactor systems, the experimental methodology differs significantly. These systems utilize pure water vapor rather than moist air, requiring precise pressure control and specialized heat exchangers. The charging process involves applying heat to the reactor while maintaining vacuum conditions to remove water vapor, which is then condensed and stored separately. During discharge, the water vapor is reintroduced to the reactive material, and the generated heat is transferred to a working fluid via heat exchangers [40]. The experimental challenges with sealed systems include maintaining perfect sealing integrity throughout thermal cycling and managing the coupling between reaction kinetics and heat exchanger efficiency.
Performance Quantification Methods
The energy storage density is calculated from the measured temperature rise and mass change of the reactive material during dehydration, accounting for both sensible and latent heat components. The thermal power output is determined from the temperature increase and flow rate of the heat transfer fluid during discharge, while the coefficient of performance (COP) is calculated as the ratio of thermal energy output during discharge to the electrical energy input for pumps, fans, and controls [41].
For both system types, scalability assessment involves operating reactors of increasing size while maintaining geometric similarity and comparing key performance metrics relative to volume and surface area. This enables identification of the dominant limiting factors—whether heat transfer, mass transport, or reaction kinetics—that emerge at larger scales [40].
Visualization of System Architectures
Thermochemical Reactor Workflow
The fundamental operational principles and material pathways of sealed and open thermochemical storage systems can be visualized through their distinct workflows:
Thermochemical Reactor Operational Workflows
The diagram illustrates the fundamental architectural differences between the systems. The sealed system operates as a closed loop where water is conserved internally between phases, while the open system continuously exchanges both mass and energy with the ambient environment throughout its operation.
Scalability Limitation Analysis
The transition from laboratory to industrial scale introduces distinct limitations for each reactor configuration, which can be systematically analyzed:
Scalability Limitation Pathways
This visualization illustrates how different technical challenges emerge as primary constraints for each system type during scale-up. Sealed systems face fundamentally different engineering hurdles compared to open systems, necessitating distinct development approaches for successful commercialization.
Research Reagent and Material Solutions
The experimental investigation and implementation of reactor systems require specific materials and reagents tailored to each configuration's operational requirements.
Table 2: Essential Research Materials for Reactor System Development
| Material/Reagent | Function | Application Context | Critical Properties |
|---|---|---|---|
| MgCl₂·6H₂O | Thermochemical storage material | Open reactor energy storage [41] | High energy density (1 GJ/m³), non-toxic, low cost |
| Zeolites | Adsorbent material | Alternative thermochemical storage | Good cyclic stability, moderate energy density |
| LiCl–CaCl₂ flux | Solvent medium | Crystal growth of advanced materials [30] | Low melting point, high solubility for precursors |
| B₂O₃ flux | Solvent medium | YCOB crystal growth [42] | High purity, suitable viscosity for crystal growth |
| Glass-to-Metal Seals | Hermetic penetration | Sealed reactor electrical feeds [43] | Thermal shock resistance, radiation stability |
| Ni-based alloys | Structural material | Corrosion resistance in aggressive media [44] | High temperature strength, corrosion resistance |
The selection of thermochemical materials represents a critical research area, with salt hydrates like MgCl₂·6H₂O offering compelling combinations of energy density, safety, and cost-effectiveness for open systems [41]. For sealed systems, specialized sealing technologies such as glass-to-metal seals provide maintenance-free integrity over extended operational lifetimes, surviving extreme temperature and pressure conditions beyond the capabilities of polymer-based alternatives [43].
In materials synthesis applications, flux growth techniques utilize specialized reagent systems like LiCl–CaCl₂ or B₂O₃ to create controlled reaction environments for crystal growth [42] [30]. These flux systems enable lower processing temperatures and improved crystal quality compared to direct solid-state reactions, though they introduce additional complexity in separating products from the flux medium—a scalability challenge in itself.
The comparative analysis reveals that neither sealed nor open reactor systems represent a universally superior approach; rather, each configuration offers distinct advantages aligned with specific application requirements and scalability pathways. Open reactor systems demonstrate compelling benefits in simplicity, reduced infrastructure requirements, and proven experimental performance with manageable scalability challenges primarily related to heat transfer management. Their ability to directly utilize ambient air as both reactant and heat transfer medium simplifies system architecture while introducing dependencies on environmental conditions that must be managed at commercial scale [40] [41].
Sealed reactor systems offer theoretical advantages in energy density and operational flexibility, particularly for applications requiring precise atmosphere control or where environmental isolation is essential. However, these systems face more complex scalability challenges related to gas transport limitations, heat exchanger design, and maintaining sealing integrity across large surface areas and through numerous penetrations [40] [43]. The development of advanced sealing technologies and optimized bed geometries represents active research areas addressing these constraints.
Future research directions should prioritize hybrid approaches that leverage the advantages of both configurations while mitigating their respective limitations. The development of standardized testing protocols across different scales will enable more direct comparison of system performance and scalability potential. Additionally, advanced materials research focusing on both thermochemical storage media and structural components will address fundamental limitations in both system types. As reactor designs evolve to meet increasing demands for sustainable energy storage and materials production, understanding these scalability challenges provides a crucial foundation for guiding both research priorities and technology development pathways.
Overcoming Synthesis Challenges: Impurity Control, Morphology, and Scalability
Identifying and Mitigating Common Defects and Impurities
Solid-state reactions are a fundamental synthesis method to obtain polycrystalline materials from solid reagents, typically requiring high temperatures to overcome kinetic barriers for ionic diffusion and product formation [1]. Within this domain, flux methods utilize a solvent medium (the flux) to facilitate crystal growth or chemical reactions at elevated temperatures. The flux acts as a liquid medium that enhances diffusion, dissolves starting materials, and promotes the crystallization of desired phases, often leading to higher purity and more well-formed crystals compared to conventional solid-state reactions [42]. This guide provides a comparative analysis of direct solid-state reaction and flux growth methods, focusing on their efficacy in controlling common defects and impurities, a critical concern for researchers and drug development professionals in the field of materials science.
Comparative Analysis of Solid-State and Flux Methods
The choice between conventional solid-state and flux-mediated synthesis significantly impacts the nature and concentration of defects in the final product. The table below summarizes a comparative analysis of their performance in managing defects and impurities.
| Aspect | Direct Solid-State Reaction | Flux Growth Method |
|---|---|---|
| Primary Mechanism | Direct diffusion and reaction between solid reagents at high temperatures [1]. | Reaction and crystallization from a solution of reactants dissolved in a molten flux solvent [42]. |
| Typical Defects | Inhomogeneous mixing, unreacted phases, irregular morphology, high porosity, grain boundary impurities [1]. | Inclusion of flux in the crystal lattice, surface imperfections, solvent occlusion [42]. |
| Common Impurities | Impurities from initial solid precursors; difficult to remove post-synthesis [1]. | Potential for flux incorporation (e.g., Li from Li(2)CO(3) flux, B from B(2)O(3) flux); often removable by washing [42]. |
| Crystal Quality / Morphology | Polycrystalline aggregates; limited control over particle size and shape [1]. | Often yields large, high-quality single crystals with well-defined facets [42]. |
| Stoichiometry Control | Challenging, especially for multi-cation phases; prone to local non-stoichiometry [1]. | Excellent for complex compositions; promotes equilibrium growth and homogeneous cation distribution [42]. |
| Mitigation Strategies | Prolonged grinding/ball milling, repeated calcination cycles, optimization of temperature/pressure [1]. | Careful flux selection, optimized cooling cycles, post-synthesis washing/etching to remove residual flux [42]. |
| Scalability | Highly scalable for large-scale polycrystalline powder production [1]. | More suitable for laboratory-scale single crystal growth; scalability can be challenging [42]. |
Key Experimental Findings and Data
- Hollow Structure Formation in Solid-State Reactions: The synthesis of LiNi({0.5})Mn({1.5})O(_4) (LNMO) hollow microspheres via solid-state reaction demonstrates how morphological defects can be engineered for performance. A mechanism analogous to the Kirkendall effect, where the outward diffusion of Mn and Ni atoms is faster than the inward diffusion of O atoms, leads to the formation of a hollow cavity [1]. This structure resulted in a high initial discharge capacity of 137.3 mAh/g at 0.1 C and excellent capacity retention of 96.5% after 200 cycles at 1 C [1].
- Flux Growth of YCOB Single Crystals: The growth of yttrium calcium oxyborate (YCOB) single crystals using a boron-tri-oxide (B(2)O(3)) self-flux successfully produced highly transparent and large single crystals [42]. Characterization by powder X-ray diffraction confirmed the formation of a single YCOB phase, while optical studies showed high transparency in the visible and UV regions (>220 nm), indicating a low concentration of optical defects [42]. This contrasts with polycrystalline materials from direct solid-state reactions, which often suffer from light scattering at grain boundaries.
Experimental Protocols for Flux Growth
The following section details a standard experimental methodology for growing single crystals via the flux method, as exemplified by the growth of YCOB [42].
Protocol: Flux Growth of YCOB Single Crystals
Objective: To synthesize high-quality yttrium calcium oxyborate (YCOB) single crystals for nonlinear optical applications using a B(2)O(3) self-flux.
Required Reagents:
- Y(2)O(3) (High Purity)
- CaCO(_3) (High Purity)
- B(2)O(3) (Flux material, High Purity)
Apparatus:
- Platinum crucible
- High-temperature furnace with programmable temperature controller
- Mortar and pestle
Step-by-Step Workflow:
Synthesis of Polycrystalline YCOB Feedstock:
- Stoichiometrically mix the high-purity powders of Y(2)O(3), CaCO(3), and B(2)O(3) in a platinum crucible according to the reaction: Y(2)O(3) + 8CaCO(3) + 3B(2)O(3) → 2YCa(4)O(BO(3))(3) + 8CO(2)↑ [42].
- Heat the mixture to 1000 °C for 24 hours to drive off carbon dioxide and initiate the solid-state reaction.
- Regrind the resulting product and heat it again to 1200 °C for 24 hours to complete the formation of a pure, homogeneous polycrystalline YCOB mass [42].
Crystal Growth:
- The synthesized YCOB powder is mixed with an excess of B(2)O(3) flux (e.g., in a 1:1 ratio by weight) and placed in a platinum crucible.
- The crucible is introduced into a high-temperature furnace and heated to a peak temperature above the saturation point (e.g., 1360 °C) to ensure complete dissolution of the YCOB feedstock in the molten flux [42].
- The melt is soaked at this temperature for several hours to achieve homogeneity.
Nucleation and Crystal Growth:
- The homogeneous solution is slowly cooled at a controlled rate (e.g., 2-4 °C per hour) through the supersaturation zone to promote spontaneous nucleation and growth of YCOB single crystals [42].
- Once the growth is complete, the temperature is lowered more rapidly to room temperature.
Post-Synthesis Processing (Separation and Cleaning):
- The solidified flux matrix, containing the grown YCOB crystals, is removed from the crucible.
- To separate the crystals, the flux is dissolved using a suitable solvent. In the case of the B(2)O(3) flux, dilute nitric acid (HNO(_3)) or hot water is used to etch away the flux material, liberating the YCOB single crystals without mechanical damage [42].
The workflow for this protocol is visualized below.
The Scientist's Toolkit: Essential Research Reagents
Successful solid-state synthesis, whether by direct or flux methods, relies on the careful selection and use of high-purity materials. The table below details key reagents and their functions in these processes.
| Reagent / Material | Primary Function | Application Context |
|---|---|---|
| Boron-tri-oxide (B₂O₃) | A self-flux and solvent; lowers the crystallization temperature and provides a medium for crystal growth [42]. | Flux growth of oxide crystals (e.g., YCOB, other borates). |
| Lithium Carbonate (Li₂CO₃) | Acts as a flux medium and can be a source of lithium ions in the synthesis of solid electrolytes [13] [42]. | Flux growth; synthesis of lithium-containing oxides (e.g., NCM cathodes, LLZO). |
| Platinum Crucible | Inert container capable of withstanding very high temperatures without contaminating the reaction melt [42]. | High-temperature synthesis (>1000 °C) of oxides, especially with basic fluxes. |
| Dolomite (CaMg(CO₃)₂) | Basic flux that reacts with and removes acidic impurities like silica (SiO₂) by forming a fluid slag [45]. | Slag formation and purification in ferrous metallurgy (iron & steel production). |
| Limestone (CaCO₃) | Decomposes to CaO, a basic flux that removes acidic impurities and forms slag [45]. | Primary flux in blast furnace operations for iron production. |
| Silica (SiO₂) | Acidic flux that combines with basic impurities (e.g., iron oxides) to form a fluid slag [45]. | Slag formation in non-ferrous metal processing (e.g., copper smelting). |
| Fluorite (CaF₂) | "Fluidity" flux; lowers the melting point and viscosity of the slag, improving impurity removal [45]. | Additive in steelmaking to enhance slag performance. |
| Borax (Na₂B₄O₇) | Lowers the melting point of metal oxides, acts as a cleaning agent and protective surface barrier [45]. | Brazing, welding, and refining of precious metals. |
| Nitric Acid (HNO₃) | Solvent for dissolving and removing residual oxide-based flux material post-crystallization [42]. | Post-synthesis cleaning of crystals grown from fluxes like B₂O₃. |
Visualizing Defect Mitigation Pathways
The strategic selection of a synthesis method initiates a specific pathway for material formation, with inherent mechanisms for defect control. The following diagram illustrates the logical decision process and the primary defect mitigation pathways associated with direct solid-state and flux growth methods.
Strategies for Controlling Particle Size and Crystallinity
Controlling particle size and crystallinity is a cornerstone of advanced materials synthesis, directly influencing the performance of materials in catalysis, electronics, and drug development. Within the broad spectrum of synthesis techniques, solid-state reaction flux methods offer unique advantages for crafting materials with precise structural characteristics. This guide objectively compares these direct methods against alternative synthesis approaches, drawing on recent experimental studies to provide a clear framework for selecting and optimizing synthesis protocols. The analysis is structured around quantifiable outcomes—primarily particle size, crystallinity, and phase purity—enabling researchers to align method selection with specific application requirements.
Comparative Analysis of Synthesis Methods
The choice of synthesis strategy profoundly impacts the structural properties of the final material. The following table provides a direct comparison of four prevalent techniques.
Table 1: Objective Comparison of Synthesis Methods for Particle Size and Crystallinity Control
| Synthesis Method | Typical Particle/Crystal Size Range | Key Crystallinity & Phase Control Factors | Reported Experimental Data |
|---|---|---|---|
| Solid-State Reaction (SSR) Flux [30] [46] | ~1 μm to large single crystals [30] [47] | • Flux composition (e.g., LiCl–CaCl₂, CsCl) [30] [46]• Heating temperature & duration [47]• Precursor powder homogeneity & density [47] | • Gd₂/₃Y₁/₃Sm₁/₃Tb₁/₃Ho₁/₃Ti₂O₂S₂: ~1 μm platy single crystals, high configurational entropy, uniform element distribution [30].• Er₂Ir₂O₇: Formation of pyrochlore phase; non-stoichiometric mixtures reduced initial oxide percentage [46]. |
| Hydrothermal/Solvothermal Synthesis [48] | Sub-micrometer to micrometer, tunable distribution [48] | • Temperature & time [48]• Structure-directing agent (e.g., morpholine) [48]• Gel composition & pH | • SAPO-34: Crystallinity up to 96.4%; Mean crystal size controlled from 180°C to 220°C; Nucleation rate: 2.84×10⁷ to 2.15×10⁹ #/s.m³; Growth rate: 2.41 to 9.06 nm/s [48]. |
| Solution-Growth with Feedback Control [6] | Millimeter to centimeter-scale single crystals [6] | • Linear growth rate (directly controlled) [6]• Solvent evaporation & supersaturation [6]• Seed crystal quality [6] | • MAPbBr₃ (Perovskite): Growth rates of 0.1-0.3 mm/h; X-ray rocking curve FWHM of 15.3 arcsec at ~0.2 mm/h [6]. |
| Flux-Regulated Crystallization (FRC) [6] | Centimeter-scale single crystals [6] | • Direct feedback control of linear growth rate [6]• Net evaporation rate via solvent infusion [6]• PID controller parameters [6] | • MAPbBr₃, CsPbBr₃: Stable growth >40 hours; High crystallinity (FWHM ~15.3 arcsec) in cm-scale crystals [6]. |
Experimental Protocols for Key Methods
Solid-State Reaction Flux Method for Pyrochlore Iridates
Protocol for Er₂Ir₂O₇ synthesis via CsCl flux, as detailed by Vlášková et al. [46]:
- Precursor Preparation: Er₂O₃ and IrO₂ oxides (99.99% purity) are dried at 200°C, weighed in a stoichiometric 1:1 ratio (Er:Ir), and thoroughly ground.
- Flax Addition: The mixed powder is combined with CsCl flux.
- Heating Process: The mixture is heated to 1000°C in a sealed quartz ampoule for 48 hours.
- Post-Synthesis Processing: The ampoule is slowly cooled to room temperature. The resulting product is washed with distilled water to dissolve the CsCl flux, leaving behind the synthesized crystals.
- Key Control Parameter: The use of a sealed ampoule and a CsCl flux counteracts the high evaporation rate and low-temperature decomposition of IrO₂, enabling the formation of the target pyrochlore phase [46].
Population Balance Modeling for Hydrothermal Crystallization
Protocol for modeling SAPO-34 crystallization kinetics, as per the study on crystallization kinetics [48]:
- Material Synthesis: SAPO-34 is hydrothermally synthesized from a gel with molar composition 1.0 Al₂O₃: 1.0 P₂O₅: 0.6 SiO₂: 4.0 morpholine: 70 H₂O at temperatures of 180, 200, and 220°C for varying times.
- Characterization: Synthesized crystals are characterized by XRD for crystallinity and crystallite size, and by FESEM/DLS for particle size distribution (PSD).
- Model Development: A population balance model (PBM) is constructed based on homogeneous nucleation and diffusion-controlled crystal growth kinetics.
- Optimization: The Grey Wolf Optimization technique is applied to solve the model equations and fit them to the experimental PSD data, enabling prediction of nucleation rate, growth rate, and final crystal size distribution [48].
Visualizing Synthesis Workflows and Control Logic
Feedback-Controlled Flux Crystallization Workflow
The flux-regulated crystallization (FRC) method represents a significant advance for achieving reproducible, high-quality crystals from solution by directly controlling the linear growth rate [6]. The following diagram illustrates its automated feedback loop.
Population Balance Modeling Logic
For hydrothermal synthesis, a population balance model (PBM) provides a theoretical framework to predict and control the crystal size distribution. The logic of applying this model is shown below [48].
The Scientist's Toolkit: Essential Research Reagents and Materials
The successful execution of synthesis protocols relies on specific reagents and materials. This table details key items for the methods discussed.
Table 2: Essential Research Reagent Solutions for Controlled Synthesis
| Reagent/Material | Function in Synthesis | Exemplary Use Case |
|---|---|---|
| Chloride Salt Fluxes (LiCl, CaCl₂, CsCl) | Low-melting solvent that facilitates atomic mobility and crystal growth at lower temperatures, improving phase purity and crystal size [30] [46]. | Growth of high-entropy oxysulfide (Gd₂/₃Y₁/₃Sm₁/₃Tb₁/₃Ho₁/₃Ti₂O₂S₂) and Er₂Ir₂O₇ pyrochlore single crystals [30] [46]. |
| Structure-Directing Agents (Morpholine) | Organic template that guides the formation of specific microporous frameworks during hydrothermal synthesis [48]. | Synthesis of SAPO-34 zeotype with CHA structure [48]. |
| High-Purity Oxide Precursors (Y₂O₃, Al₂O₃) | Raw materials for solid-state reactions; their purity, particle size, and mixing homogeneity are critical for achieving monophasic, high-density products [47]. | Solid-state synthesis of Y₃Al₅O₁₂ (YAG) garnet for laser and scintillator applications [47]. |
| PID-Controlled Syringe Pump | Actuator in a feedback loop that precisely infuses solvent to regulate net evaporation rate, enabling direct control of linear crystal growth rate [6]. | Flux-regulated crystallization (FRC) of MAPbBr₃ and CsPbBr₃ perovskite single crystals [6]. |
Optimizing Thermodynamic Stability and Composition in Multicomponent Ceramics
The development of advanced multicomponent ceramics represents a frontier in materials science, driven by demands for improved performance in extreme environments. These materials, including high-entropy ceramics (HECs) and multiphase multicomponent ultrahigh-temperature ceramics (UHTCs), offer unprecedented opportunities for tailoring mechanical, chemical, and thermal properties for applications ranging from aerospace propulsion systems and nuclear reactors to high-performance cutting tools and thermal protection coatings [49] [50]. However, a significant challenge in advancing their discovery lies in accurately predicting their single-phase stability and formation ability, which directly influences their thermodynamic stability and ultimate performance characteristics [49].
Two primary methodological frameworks have emerged for optimizing these materials: computational prediction approaches, particularly ab initio free energy modeling, and direct solid-state reaction methods, including flux-assisted techniques. Computational methods enable rapid screening of vast compositional spaces to identify promising candidates with enhanced thermodynamic stability, while solid-state reactions provide the experimental pathway to synthesize and validate these materials. The synergy between these approaches—where computational predictions guide experimental synthesis—represents the cutting edge of ceramic materials design [49] [51]. This guide objectively compares these methodologies, their performance outcomes, and the experimental protocols that underpin advances in multicomponent ceramic optimization.
Comparative Analysis of Stability Prediction Methods
Computational Free Energy Modeling
The thermodynamic stability of multicomponent systems is fundamentally governed by Gibbs free energy (ΔG = ΔH - TΔS), where the most stable phase possesses the lowest free energy [49]. Computational approaches have evolved from descriptor-based predictions to direct free energy calculations using ab initio methods. Descriptor-based approaches, including Entropy Forming Ability (EFA) and the Disordered Enthalpy-Entropy Descriptor (DEED), correlate specific material properties with phase stability but rely heavily on empirical correlations with existing experimental data [49]. In contrast, ab initio free energy modeling directly calculates free energy terms, providing a more physics-based approach to stability prediction.
Key Advantages: Ab initio free energy modeling demonstrates superior computational efficiency and physical rigor compared to descriptor-based methods. This approach has successfully predicted phase stability in high-entropy borides (HEBs) and carbides (HECs), with validation against existing experimental data and correction of previous literature inaccuracies [49]. The methodology enables researchers to screen potential ceramic compositions without expensive trial-and-error experimentation, significantly accelerating materials discovery.
Performance Data: In direct comparisons, the ab initio free energy model accurately differentiated between single-phase and multi-phase formations in quinary high-entropy ceramic systems. When a discrepancy appeared between predictions and literature for a specific composition, experimental synthesis confirmed the accuracy of the ab initio model, demonstrating its predictive reliability [49]. The method also identified several new single-phase compositions not previously reported, highlighting its discovery potential.
Direct Solid-State Reaction and Flux Growth Methods
Solid-state reaction methods represent the conventional experimental approach for synthesizing multicomponent ceramics. This technique involves heating powder precursors to elevated temperatures under controlled atmospheric conditions, facilitating interdiffusion and reaction between solid components [51]. Within this category, flux crystal growth serves as a specialized technique where crystals grow from a high-temperature molten medium (the flux), which enhances diffusion and reaction kinetics while potentially lowering synthesis temperatures [52].
Key Advantages: Flux methods offer enhanced control over crystal growth, enabling the production of high-quality single crystals suitable for characterizing anisotropic physical properties [52]. The flux medium provides improved mass transport and can moderate reaction rates, leading to more thermodynamically stable phases with fewer defects. These methods have proven particularly valuable for systems containing volatile components, such as phosphides and arsenides [52].
Performance Data: Experimental studies demonstrate that flux growth methods successfully produce millimeter-sized single crystals of complex multinary phosphides and arsenides, which are difficult to synthesize through conventional solid-state reactions [52]. The quality of these crystals enables precise characterization of structure-property relationships, facilitating optimization of functional characteristics.
Table 1: Comparison of Stability Optimization Methods for Multicomponent Ceramics
| Method Characteristic | Computational Free Energy Modeling | Direct Solid-State Reaction | Flux-Assisted Synthesis |
|---|---|---|---|
| Primary Function | Predict phase stability from first principles | Synthesize ceramics from powder precursors | Grow high-quality single crystals |
| Throughput | High (rapid screening of compositions) | Moderate (batch processing) | Low (requires optimization) |
| Key Output | Predicted phase stability & formation ability | Bulk polycrystalline ceramics | Single crystals for characterization |
| Temperature Range | Not applicable | High temperatures (often >1500°C) | Variable (dependent on flux system) |
| Material Systems | HEBs, HECs, nitrides, oxides | Broad applicability | Phosphides, arsenides, complex intermetallics |
| Experimental Validation | Required for confirmation | Direct material production | Direct material production |
| Limitations | Dependent on database completeness & model accuracy | Kinetic limitations, incomplete reactions | Flux incorporation, removal challenges |
Experimental Protocols and Workflows
Computational Prediction Workflow
The computational prediction of phase stability in multicomponent ceramics follows a systematic workflow that integrates first-principles calculations with thermodynamic modeling. The process begins with defining the target composition system, typically equimolar quinary systems for high-entropy ceramics [49]. Special quasirandom structures (SQS) are generated to model the disordered configurations using Monte Carlo methods, creating supercells where metal lattice sites are occupied by different elements [49].
First-principles calculations within the density functional theory (DFT) framework form the core of the methodology. These computations employ the Vienna ab initio simulation package (VASP) with the Perdew-Burke-Ernzerhof (PBE) exchange-correlation functional and projector-augmented-wave potentials [49]. The conjugate gradient method optimizes SQS structures with careful parameterization, including a plane-wave cut-off energy of 520 eV and energy tolerance of 0.002 meV/atom [49].
The thermodynamic analysis calculates the formation enthalpy (ΔH) with respect to the most stable competing phases identified from databases like the Materials Project, including elemental, binary, and ternary compounds [49]. The configurational entropy (ΔS) assumes ideal mixing, while vibrational entropy contributions can be estimated using machine learning methods [53] [51]. Phase stability is then evaluated by comparing the Gibbs free energy of the disordered high-entropy phase against competing phases across relevant temperature ranges [49].
Diagram 1: Computational stability prediction workflow. This diagram illustrates the integrated computational materials engineering (ICME) approach for predicting phase stability in multicomponent ceramics, combining first-principles calculations with thermodynamic modeling.
Solid-State Reaction Synthesis Protocol
The experimental synthesis of multicomponent ceramics via solid-state reaction follows a multi-stage process focused on achieving phase-pure materials with controlled microstructure. For high-entropy carbide systems such as (TiZrHfMoW)C, the protocol begins with precursor preparation using binary carbide powders (TiC, ZrC, HfC, Mo₂C), tungsten powders, and graphite powders, all with purity >99% [49]. These precursors are weighed in stoichiometric proportions and subjected to high-energy ball milling using silicon nitride milling media in an ethanol medium. The milling sequence typically follows intervals of 5 minutes of milling followed by 5 minutes of rest to prevent overheating, continuing for a total of 2 hours [49].
The mixed powders are then dried at 65°C before consolidation. Arc-melting represents a common consolidation method, performed in an inert argon atmosphere with cooling on a water-cooled copper hearth [49]. For bulk samples, the consolidated material may be cut using a wafering blade and mirror-polished on one side using diamond lapping film discs. A critical homogenization step follows, involving thermal annealing of arc-melted samples for 12 hours in a high vacuum (10⁻⁵ Pa) at 1650°C [49]. Furnace cooling at a controlled rate of 300°C/minute completes the thermal processing.
Characterization protocols to verify phase formation and microstructure include X-ray diffraction (XRD) analysis on polished surfaces using equipment such as a Bruker D8 discovery X-ray diffractometer with a two-dimensional detector [49]. Standard XRD parameters include a scan range (2θ) of 20°–80° with a step of 20° and acquisition times of at least 120 seconds per step. Supplementary characterization often involves scanning electron microscopy (SEM) for microstructural analysis and energy-dispersive X-ray spectroscopy (EDS) for compositional mapping [49].
Flux-Assisted Synthesis Methodology
Flux-assisted synthesis employs a high-temperature molten medium to facilitate crystal growth of complex ceramic compounds, particularly valuable for systems with volatile components or challenging crystal structures. The protocol begins with flux selection based on multiple criteria, including solubility parameters, chemical inertness, melting temperature, and removal considerations [52]. For phosphides and arsenides, common flux materials include tin (Sn), bismuth (Bi), lead (Pb), and aluminum (Al), each offering specific advantages for different material systems [52].
The process involves combining precursor elements with the flux material in sealed containers, often quartz ampoules, to control volatility and prevent oxidation. Typical flux-to-precursor ratios range from 10:1 to 20:1, ensuring sufficient solvent volume for crystal growth [52]. The reaction vessel undergoes a carefully controlled thermal profile with heating above the flux melting point to dissolve reactants, followed by slow cooling to promote controlled crystallization. Cooling rates as slow as 0.4 K/hour may be employed, requiring weeks to complete crystal growth for challenging systems [52].
Post-synthesis processing involves flux removal through multiple potential methods: mechanical separation and crystal polishing, high-temperature centrifugation of liquid flux, or chemical dissolution using appropriate solvents [52]. For metallic fluxes like Sn or Pb, acid treatments (HCl, sometimes with H₂O₂/acetic acid) effectively remove the flux matrix, while salt fluxes (KCl/NaCl) dissolve in water or ethanol. The specific removal method depends on the chemical stability of the target crystals in the dissolution media.
Performance Comparison Across Ceramic Systems
High-Entropy Carbides and Borides
High-entropy carbides and borides represent extensively studied ceramic systems where both computational and experimental methods have demonstrated significant success in optimizing thermodynamic stability. Performance comparisons reveal distinct advantages for integrated approaches that combine computational screening with experimental validation.
Table 2: Performance Data for High-Entropy Carbide and Boride Systems
| Material System | Synthesis Method | Key Findings | Stability Outcome |
|---|---|---|---|
| Quinary HECs | Arc-melting & annealing (1650°C, 12h) | Single-phase rock-salt structure confirmed by XRD | High thermodynamic stability maintained after homogenization |
| Quinary HEBs | Computational prediction followed by validation | Ab initio free energy model correctly identified single-phase formers | Accurate prediction of phase stability without prior experimental data |
| Multi-phase Multicomponent Carbide (Hf-Zr-Ti-Ta-C) | Solid-state reaction | Three-phase structure (Hf-rich, Ta-rich, Ti-rich carbides) enhanced ablation resistance | Thermodynamic-driven solid-state diffusion during ablation improved stability |
For high-entropy carbides, experimental results confirm that computational free energy modeling accurately predicts single-phase stability across multiple quinary systems [49]. The ab initio approach successfully identified new single-phase compositions beyond those previously reported in literature, demonstrating its discovery potential. Experimental validation through synthesis and characterization confirmed these predictions, with XRD analysis verifying single-phase formation [49].
In multiphase multicomponent carbide systems such as Hf-Zr-Ti-Ta-C, thermodynamic-driven solid-state diffusion processes during high-temperature ablation significantly enhance performance [50]. This system forms three distinct carbide phases (Hf-rich, Ta-rich, and Ti-rich) that undergo interdiffusion of metal ions (primarily Zr and Ti) during ablation, leading to Zr- and Ti-rich oxide formation [50]. The resulting Ta enrichment in the residual carbide phase improves thermodynamic stability at relatively high oxygen partial pressures, demonstrating how in situ phase transformations can optimize stability under service conditions.
Multicomponent Nitride Systems
Multicomponent nitride systems showcase remarkable property optimization through careful composition control, particularly demonstrating anomalous thermal conductivity behavior that challenges conventional ceramic characteristics. Research on aluminum nitride (AlN)-based multicomponent nitrides with diverse metallic elements reveals how composition-induced lattice distortion and conductive network formation transform thermal transport properties [54].
The TiAlN system achieves high room-temperature thermal conductivity of 48.38 W·m⁻¹·K⁻¹, while increased diversity of metallic elements induces severe lattice distortion that suppresses phonon thermal conduction [54]. Counterintuitively, these multicomponent systems simultaneously form metallic nitride conductive networks that significantly increase electronic thermal conductivity. This synergistic electron-phonon regulation successfully transforms the thermal conductivity-temperature (κ-T) dependence from negative (typical for ceramics) to positive (typical for metals) [54].
The TiZrVCrAlN composition demonstrates exceptional performance with a linear 112% thermal conductivity increase from 8.65 W·m⁻¹·K⁻¹ at -60°C to 18.34 W·m⁻¹·K⁻¹ at 900°C [54]. This represents the highest reported performance among known positive-κ ceramics in both operating temperature range and conductivity values. Moreover, the material maintains robust mechanical integrity with 24.5 GPa hardness and 273 MPa bending strength, illustrating successful optimization of both thermodynamic stability and functional properties [54].
Oxide Ceramic Systems
Multicomponent oxide ceramics benefit from both conventional solid-state reactions and flux-assisted methods, with performance optimization closely tied to processing conditions and phase formation. Studies on multicomponent ceramics based on oxide compounds of cerium, tellurium, bismuth, tungsten, and zinc demonstrate how thermal sintering conditions control phase transformation processes and resultant properties [55].
Annealing temperature elevation above 1000°C promotes zinc tungstate (ZnWO₄) inclusion growth through recrystallization of ZnTeO₃ and Bi₂WO₆ phases [55]. These microstructural developments create glass-like inclusions with reinforcing effects, enhancing ceramic resistance to external influences including high-temperature destruction and hydrogenation processes. Performance analysis confirms that zinc tungstate inclusion dominance improves resistance to both hydrogenation-induced destruction and thermal aging, addressing key destabilization mechanisms that typically degrade ceramic strength properties and thermal insulation performance [55].
Research Reagent Solutions
Table 3: Essential Research Reagents for Multicomponent Ceramic Synthesis
| Reagent Category | Specific Examples | Function in Synthesis | Application Notes |
|---|---|---|---|
| Precursor Powders | Binary carbides (TiC, ZrC, HfC, Mo₂C), tungsten powder, graphite | Provide elemental constituents for final ceramic phase | Purity >99% essential to avoid impurity phases |
| Metallic Fluxes | Sn, Bi, Pb, Al, Ga, In | High-temperature solvent for crystal growth; enhances diffusion | Selected based on solubility parameters and chemical inertness |
| Salt Fluxes | KCl, NaCl | Low-temperature flux for oxygen-sensitive materials | Water-soluble for easy removal after synthesis |
| Milling Media | Si₃N₄ balls, jars | High-energy mechanical alloying of precursors | Ethanol medium prevents oxidation during milling |
| Reaction Atmospheres | Argon, nitrogen, oxygen | Control oxidation state and prevent contamination | Selection depends on material system reactivity |
| Characterization Standards | Silicon standard for XRD calibration | Ensure accurate phase identification | Critical for quantitative phase analysis |
The selection of appropriate research reagents fundamentally influences the success of multicomponent ceramic synthesis and optimization. High-purity precursor materials (>99%) prove essential for achieving phase-pure products, as impurities can catalyze undesirable phase formation or inhibit diffusion processes [49] [50]. Metallic fluxes require careful matching to the target material system, with Sn flux preferred for phosphides due to higher phosphorus solubility, while Bi flux works effectively for arsenides [52]. Salt fluxes like KCl/NaCl offer advantages for oxygen-sensitive materials but require water solubility for practical removal after synthesis.
Milling media selection impacts both contamination risk and processing efficiency. Silicon nitride (Si₃N₄) milling media provides excellent wear resistance with minimal contamination compared to traditional tungsten carbide media, which can introduce foreign elements [49]. Reaction atmosphere control represents another critical parameter, with argon appropriate for most carbide systems, while nitrogen atmospheres enable nitride formation [49] [54]. Each reagent category contributes specifically to controlling reaction pathways, diffusion kinetics, and ultimate phase stability in the synthesized multicomponent ceramics.
The optimization of thermodynamic stability and composition in multicomponent ceramics increasingly relies on integrated methodologies that combine computational prediction with experimental validation. Ab initio free energy modeling has demonstrated remarkable accuracy in predicting phase stability across high-entropy boride, carbide, and nitride systems, significantly accelerating materials discovery while reducing reliance on empirical trial-and-error approaches [49]. Meanwhile, advanced synthesis techniques including optimized solid-state reactions and flux-assisted methods continue to provide essential experimental validation and enable the production of materials with enhanced properties.
Future developments will likely focus on several key areas: improved modeling of configurational disorder and its entropy contributions at elevated temperatures, development of more sophisticated kinetic models that account for diffusion limitations in solid-state reactions, and enhanced integration of computational predictions with autonomous synthesis platforms for rapid experimental validation [51]. The continuing expansion of computational databases, coupled with advanced in situ characterization techniques, will further bridge the gap between predicted and realized material structures. For researchers navigating this complex landscape, the strategic combination of computational screening with carefully optimized synthesis protocols represents the most promising path toward designing multicomponent ceramics with precisely tailored stability and performance characteristics for extreme environment applications.
Solid-state reactions represent a cornerstone technique in physics, chemistry, and material science for synthesizing compounds, prized primarily for their simplicity in scaling operations up or down [56]. Despite this significant advantage, these methods confront substantial limitations that can impede research progress and practical applications. The core challenges include inherently long reaction durations, requirements for high temperatures, and the frequent necessity for extensive post-synthesis purification to achieve desired material properties. These limitations stem from the fundamental nature of solid-state reactions, which often proceed through direct atomic diffusion in the solid phase, leading to uneven chemical reactions that result in wide variances in optical and microstructural properties [56]. For instance, research on LaCe₀.₉Th₀.₁CuOy systems has demonstrated only approximately 72% homogeneity, with the remaining 28% exhibiting heterogeneity—an anomaly that may underlie unresolved mechanisms in various solid-state devices [56].
Within the broader context of direct solid-state reaction flux methods research, controlling reaction pathways and outcomes remains a persistent challenge. The inability to precisely steer reaction flux often necessitates subsequent purification steps or results in inefficient synthesis, particularly when targeting specific product distributions or material characteristics. Similar purification challenges exist in other synthetic fields, such as oligonucleotide synthesis, where imperfect coupling efficiencies (typically around 99% per step) generate truncated failure sequences and deletion byproducts that must be removed through various purification techniques [57] [58]. This article provides a comprehensive comparison of approaches addressing these limitations across different synthetic domains, with particular emphasis on experimental data and methodologies relevant to researchers seeking to optimize their synthetic strategies.
Comparative Analysis of Methodologies and Performance Data
Solid-State Reaction Limitations and Characterization Data
The intrinsic limitations of solid-state synthesis techniques manifest clearly in experimental studies of material systems. Research on LaCeTh₀.₁CuOy polycrystalline growth via solid-state reaction reveals fundamental challenges through comprehensive characterization data. The table below summarizes key experimental findings from this investigation:
Table 1: Experimental Characterization Data for LaCeTh₀.₁CuOy Solid-State Synthesis
| Characterization Method | Key Findings | Implications for Limitations |
|---|---|---|
| Scanning Electron Microscopy (SEM) | Revealed surface morphology influenced predominantly by copper, oxygen, and cerium | Demonstrates elemental heterogeneity issues |
| Transition Electron Microscopy (TEM) | Provided bulk microstructure analysis | Confirms inhomogeneity at nanoscale |
| X-ray Diffractometry (XRD) | Identified unique combination of phases yielding new electronic properties | Indicates complex phase formation challenges |
| Homogeneity Analysis | Approximately 72% homogeneity, 28% heterogeneity | Quantifies inherent material inconsistency |
| Structural Impact Ratio | Inner to outer CuO₂ planes ratio of 5:1 | Suggests anisotropic reaction progress |
This data collectively illustrates how uneven chemical reactions during solid-state synthesis translate directly into wide variances in optical and microstructural properties [56]. The observed 28% heterogeneity represents a significant limitation for applications requiring precise material properties, potentially explaining performance inconsistencies in solid-state devices. The identification of a 5:1 structural impact ratio between inner and outer CuO₂ planes further highlights the anisotropic nature of these reactions, suggesting fundamental challenges in achieving uniform reaction progress throughout material systems [56].
Advanced Solid-State Approaches: Fluid-Induced Solid-State Diffusion
Recent investigations into fluid-induced solid-state diffusion (FI-SSD) mechanisms demonstrate promising approaches for addressing traditional solid-state synthesis limitations. Research on cobalt-bearing pyrite from Cu-Co ore deposits has revealed two distinct kinetic mechanisms for element mobility, characterized through advanced microanalytical techniques:
Table 2: Comparison of Diffusion Mechanisms in Sulfide Mineral Systems
| Diffusion Mechanism | Concentration Profile Characteristics | Fit Statistics (R²) | Element Transfer Rates | Key Features |
|---|---|---|---|---|
| Transient-State Diffusion | Time-evolving concentrations, typical error function shape | 0.89 | Standard lattice diffusion rates | Traditional model, slower process |
| Steady-State Diffusion with Concentration-Dependent Diffusivity | Constant flux, sharp decline followed by gradual decrease | 0.97 | ~2 orders of magnitude higher than vacancy-mediated lattice diffusion | Maintains chemical gradient, vacancy-facilitated |
This research demonstrates that steady-state diffusion with concentration-dependent diffusivities, facilitated by vacancy pathways in the presence of fluids, provides significantly better fit to observed concentration profiles (R² = 0.97 versus 0.89 for transient-state model) [59]. The key advantage of this mechanism lies in its dramatically enhanced efficiency, with element transfer rates nearly two orders of magnitude higher than traditional vacancy-mediated lattice diffusion [59]. This approach effectively maintains a chemical gradient between the mineral surface and contacting fluid, thereby sustaining high diffusion rates and potentially addressing the traditional limitations of long durations and high temperatures in solid-state synthesis.
Oligonucleotide Synthesis Purification Methods
While operating in a different domain, oligonucleotide synthesis faces analogous challenges with synthesis imperfections requiring post-synthesis purification. The stepwise addition of nucleotides with approximately 99% coupling efficiency per step inevitably generates failure sequences that must be removed through various purification techniques [57] [58]. The following table provides a comparative analysis of common purification methods:
Table 3: Performance Comparison of Oligonucleotide Purification Methods
| Purification Method | Purity Range | Yield | Optimal Length Range | Key Applications | Limitations |
|---|---|---|---|---|---|
| Desalting | Not applicable (doesn't remove failure sequences) | High | 5-100 bp | PCR, sequencing, microarrays | Doesn't remove truncated sequences or deletions |
| Reverse-Phase Cartridge (RPC) | ~70-80% | Moderate (≥80%) | 7-55 bp | Fluorescent sequencing, gel shift assays | Doesn't remove internal deletions; limited scale |
| HPLC | >85-90% | Medium | 10-55 bp | cDNA library construction, cloning, mutagenesis, NGS | Resolution decreases with length; secondary structures problematic |
| PAGE | >90-95% | Low | 7-100 bp | Cloning, mutagenesis, gel shift assays, NGS | Low yield; incompatible with some modifications |
| Dual HPLC | ~90% | Medium | Varies | High-purity applications (qPCR, therapeutics) | High cost; complex operation |
The data reveals the inherent tradeoff between purity and yield across purification methods, with PAGE purification achieving the highest purity (>90%) but at the cost of significantly reduced final yield [57] [58] [60]. HPLC methods strike a balance with >85% purity and medium yield, making them suitable for various applications including those requiring modified oligonucleotides [57] [58]. The length-dependent performance degradation across most methods highlights how synthesis limitations become more pronounced with increasing molecular complexity.
Experimental Protocols and Methodologies
Protocol for Analyzing Solid-State Reaction Homogeneity
Objective: To quantitatively assess homogeneity and structural properties of solid-state synthesized materials.
Materials and Reagents:
- Polycrystalline sample (e.g., LaCeTh₀.₁CuOy)
- Scanning Electron Microscope (SEM)
- Transition Electron Microscope (TEM)
- X-ray Diffractometer (XRD)
- Sample preparation equipment (polishing, coating)
Procedure:
- Prepare polycrystalline sample via solid-state reaction protocol with appropriate stoichiometric calculations and mixing.
- Subject the synthesized material to SEM analysis at multiple magnification levels to assess surface morphology and elemental distribution.
- Prepare thin sections for TEM analysis to investigate bulk microstructure properties.
- Conduct XRD analysis to identify phase composition and crystallographic structure.
- Perform quantitative image analysis on SEM/TEM data to calculate homogeneity percentage.
- Analyze coordination environments of constituent elements, particularly focusing on CuO₂ planes.
- Calculate structural impact ratios based on coordination number differences between inner and outer planes.
Data Analysis: Calculate homogeneity percentage by dividing homogeneous area by total area analyzed. Determine elemental influence on morphology through correlation analysis. Compute structural impact ratios from coordination number data [56].
Protocol for Studying Fluid-Induced Solid-State Diffusion
Objective: To characterize diffusion kinetics and mechanisms in mineral systems using advanced analytical techniques.
Materials and Reagents:
- Co-bearing pyrite samples
- High-resolution Electron Probe Microanalyzer (EPMA)
- Electron Backscatter Diffraction (EBSD) system
- Atom Probe Tomography (APT) instrument
- Aberration-corrected Scanning Transmission Electron Microscope (AC-STEM)
- Diffusion modeling software
Procedure:
- Collect and characterize Co-rich pyrite crystals from appropriate geological formations.
- Perform backscattered electron (BSE) imaging and micro X-ray fluorescence (XRF) mapping to identify Co distribution patterns.
- Conduct EPMA spot analyses to determine Co and As concentrations across defined zones.
- Perform quantitative crystallographic mapping using EBSD to assess deformation and misorientation.
- Prepare APT specimens from boundary interfaces between Co-enriched and Co-depleted zones.
- Acquire concentration profiles across boundaries using high-resolution EPMA maps.
- Model concentration profiles using both transient-state and steady-state diffusion models.
- Calculate diffusivity ratios (D₂/D₁) from corrected diffusion profiles.
- Validate findings through first-principles calculations and STEM simulations.
Data Analysis: Fit concentration profiles to both transient-state and steady-state diffusion models. Compare fit statistics (R² values) to determine superior model. Calculate corrected diffusivity ratios accounting for interface angles. Quantify vacancy densities and their relationship to diffusion fluxes [59].
Protocol for Oligonucleotide Purification Selection and Validation
Objective: To select and implement appropriate purification methods based on oligonucleotide characteristics and application requirements.
Materials and Reagents:
- Crude oligonucleotide synthesis product
- Appropriate purification equipment (HPLC system, PAGE apparatus, or cartridge)
- Desalting columns (G-25 or G-50)
- Elution buffers
- Mass spectrometry or capillary electrophoresis equipment for quality control
Procedure:
- Determine oligonucleotide parameters: length, modification presence, downstream application.
- Select purification method based on application requirements:
- For PCR or sequencing: Choose desalting
- For cloning or mutagenesis: Choose PAGE or HPLC
- For modified oligonucleotides: Choose HPLC
- For highest purity applications: Choose PAGE or Dual HPLC
- Execute chosen purification protocol:
- Desalting: Use precipitation or sizing resins to remove small molecules
- Cartridge: Employ hydrophobic matrix for separation, elute full-length product
- HPLC: Utilize reversed-phase or anion exchange chromatography for high-resolution separation
- PAGE: Implement denaturing polyacrylamide gel electrophoresis for length-based separation
- Desalt and concentrate purified oligonucleotide as needed.
- Verify purity and identity through mass spectrometry or capillary electrophoresis.
Data Analysis: Calculate yield percentage based on initial crude product. Determine purity through analytical methods. Verify removal of failure sequences through appropriate analytical techniques [57] [58] [60].
Research Reagent Solutions Toolkit
Table 4: Essential Research Reagents and Materials for Solid-State and Purification Studies
| Item | Function | Application Context |
|---|---|---|
| Scanning Electron Microscope (SEM) | Surface morphology and elemental distribution analysis | Solid-state reaction characterization |
| Transition Electron Microscope (TEM) | Bulk microstructure investigation | Nanoscale homogeneity assessment |
| X-ray Diffractometer (XRD) | Phase identification and crystallographic structure determination | Solid-state reaction product characterization |
| Electron Probe Microanalyzer (EPMA) | High-resolution elemental concentration mapping | Diffusion profile measurement in mineral systems |
| Atom Probe Tomography (APT) | Atomic-scale 3D compositional analysis | Interface characterization in diffusion studies |
| HPLC System with Reversed-Phase Columns | High-resolution separation based on hydrophobicity | Oligonucleotide purification |
| Denaturing PAGE Apparatus | Length-based separation with single-base resolution | High-purity oligonucleotide purification |
| Anion Exchange HPLC Columns | Charge-based separation of biomolecules | Purification of oligonucleotides with secondary structures |
| Desalting Columns (G-25/G-50) | Removal of small molecule contaminants | Rapid cleanup of oligonucleotides for standard applications |
Visualization of Experimental Workflows and Relationships
Solid-State Synthesis and Characterization Workflow
Solid-State Synthesis and Characterization Workflow
Oligonucleotide Purification Selection Logic
Oligonucleotide Purification Selection Logic
The comprehensive analysis presented herein demonstrates that limitations surrounding long durations, high temperatures, and post-synthesis purification represent significant yet addressable challenges across multiple synthetic domains. In solid-state reactions, the inherent 28% heterogeneity observed in model systems [56] underscores the fundamental materials-level obstacles that necessitate either improved synthetic control or compensatory purification strategies. The emerging approach of fluid-induced solid-state diffusion, with its demonstrated ability to enhance element transfer rates by nearly two orders of magnitude through vacancy-facilitated, steady-state mechanisms [59], offers promising avenues for addressing the traditional limitations of solid-state synthesis.
The parallel challenges in oligonucleotide synthesis, where stepwise coupling efficiencies inevitably generate failure sequences requiring removal [57] [58], highlight the universal nature of synthetic imperfection across chemical domains. The methodological insights gleaned from both fields—whether through vacancy-accelerated diffusion in minerals or chromatography-based separation of biomolecules—provide complementary approaches for managing the inescapable tradeoffs between synthetic efficiency, product purity, and process duration. These comparative perspectives enrich our fundamental understanding of reaction flux control and provide researchers with diverse strategic options for optimizing their specific synthetic challenges, ultimately advancing capabilities across materials science, chemistry, and drug development domains.
Leveraging Mechanochemical Activation and Alternative Sulfurization Reagents
Solid-state synthesis is undergoing a transformative shift with the advancement of mechanochemical activation and specialized sulfurization reagents, enabling researchers to overcome traditional limitations in chemical reactivity and synthetic efficiency. These methodologies provide powerful alternatives to conventional solution-based approaches, offering enhanced reaction kinetics, reduced environmental impact, and access to novel chemical spaces. Within pharmaceutical development and materials science, the strategic application of mechanochemistry and sophisticated sulfur-transfer agents is opening new pathways for constructing complex molecular architectures, particularly sulfur-containing compounds with therapeutic potential. This guide provides a systematic comparison of these emerging technologies, offering experimental data and protocols to inform research decisions within the broader context of solid-state reaction flux optimization.
Mechanochemical Activation: Principles and Performance
Fundamental Mechanisms and Comparative Performance
Mechanochemical activation utilizes mechanical energy to induce chemical and physical transformations in solid-state systems, operating through several distinct mechanisms. Mechanical activation (MA) primarily creates microstructural changes through surface area increase, crystallite size reduction, and microstrain induction without chemical reactions. In contrast, mechanochemical activation (MCA) involves co-milling the target material with another reactant to promote partial chemical reactions and enhance reactivity beyond what MA alone can achieve [61].
The performance differential between these approaches is significant. In the leaching of natural chalcopyrite (CuFeS₂), MCA with iron powder for 30 minutes increased copper extraction to 95.2%, substantially outperforming MA alone, which reached only 67.8% extraction under identical conditions and timeframes [61]. This represents an approximately 40% enhancement in process efficiency through the MCA approach.
Table 1: Performance Comparison of MA versus MCA in Chalcopyrite Processing
| Activation Method | Activation Time | Additive | Cu Extraction (%) | Relative Improvement |
|---|---|---|---|---|
| None (Reference) | - | - | ~15% | - |
| Mechanical (MA) | 30 minutes | None | 67.8% | 4.5x |
| Mechanochemical (MCA) | 30 minutes | Fe powder | 95.2% | 6.3x |
Beyond metallurgical processing, mechanochemistry demonstrates remarkable efficacy in environmental applications. For vapor-phase mercury immobilization, pyrrhotite (Fe₇S₈) activated with CuCl₂ via mechanochemical grinding achieved a Hg⁰ adsorption capacity of 283.3 mg·g⁻¹ – approximately 400 times greater than raw pyrrhotite and surpassing many synthetic nanostructured metal sulfides [62]. This dramatic enhancement stems from created lattice defects, increased surface area, and generation of additional mercury-philic active sites through the mechanochemical process.
Kinetic Enhancement Mechanisms
The dramatic rate enhancements observed in mechanochemical systems can be understood through recent theoretical advances. A scaling theory developed by Yamamoto et al. explains that mechanical force-induced convective flows in the product-rich phase between reactant particles significantly enhance reaction rates by reducing the diffusion layer thickness [63]. This theory predicts that for diffusion-limited reactions, convective flows accelerate the process by increasing reactant concentration gradients, while for reaction-limited systems, the convective flows have minimal effect on kinetics [63].
The presence of small amounts of solvent as a plasticizer further enhances these convective flows, explaining why liquid-assisted grinding often dramatically improves mechanochemical reaction rates [63]. This theoretical framework provides a foundation for optimizing milling conditions, additives, and reaction parameters to maximize kinetic advantages across different chemical systems.
Experimental Protocol: Mechanochemical Activation of Sulfide Minerals
Materials: Natural pyrite concentrate (FeS₂), nitrogen gas, copper(II) chloride dihydrate (CuCl₂·2H₂O), agate mortar and pestle [62].
Equipment: Tube furnace, planetary ball mill, sieves (200 mesh), X-ray diffractometer for characterization [62] [61].
Procedure:
- Calcination: Place natural pyrite in tube furnace. Heat to 600°C under nitrogen atmosphere for 2 hours to convert to magnetic pyrrhotite (Fe₇S₈) [62].
- Mechanochemical Modification: Combine resulting Fe₇S₈ with CuCl₂·2H₂O powder in 1:1 mass ratio. Grind mixture in agate mortar for 5 minutes under ambient conditions [62].
- Post-processing: Dry activated product at 100°C for 12 hours. Sieve through 200-mesh screen for particle size uniformity [62].
- Characterization: Analyze phase composition by XRD, examine surface morphology by SEM, and determine specific surface area by BET analysis [62] [61].
Key Processing Parameters:
- Mechanical activation: Planetary ball mill, 300 rpm, 5-60 minutes, ball-to-powder ratio 20:1 [61].
- Mechanochemical activation: Co-mill with Fe or Fe₂O₃ (10-20 wt%) under identical milling conditions [61].
Diagram 1: Mechanochemical activation workflow for enhanced adsorption
Sulfurization Reagents: Comparative Analysis and Applications
Performance Comparison of Sulfur Transfer Reagents
Sulfurization reagents facilitate the critical introduction of sulfur atoms into organic molecules and materials, with significant implications for pharmaceutical development, polymer science, and materials engineering. The performance of these reagents varies considerably based on their molecular structure, reaction kinetics, and compatibility with different substrate classes.
Table 2: Comparison of Sulfurization Reagents in Oligonucleotide Synthesis
| Reagent | Concentration | Optimal Sulfurization Time | Coupling Efficiency | Stability in Solution |
|---|---|---|---|---|
| Beaucage Reagent | 0.05 M | 4 minutes | ≥98% (after 4 min) | Limited stability |
| DDTT (Sulfurizing Reagent II) | 0.03 M | 4 minutes | ≥98% (after 4 min) | Improved stability |
| DDTT (Sulfurizing Reagent II) | 0.05 M | 1-4 minutes | ≥98% (after 1 min) | Improved stability |
| Pentanidium-Catalyzed | Catalytic | Not specified | High enantioselectivity | Not specified |
For oligonucleotide synthesis incorporating phosphorothioate linkages, DDTT (Sulfurizing Reagent II) demonstrates superior performance compared to traditional Beaucage Reagent, achieving ≥98% coupling efficiency in just 1 minute versus 4 minutes required for Beaucage Reagent at equivalent concentrations [64]. This four-fold reduction in processing time significantly enhances synthetic throughput while maintaining excellent yield and purity.
In pharmaceutical contexts, sulfur monochloride (S₂Cl₂) serves as a versatile reagent for introducing sulfur groups into drug molecules, enhancing biological activity in antibiotics and antifungal agents [65]. Its reactivity enables more efficient synthesis routes, reducing costs and improving yields in active pharmaceutical ingredient (API) manufacturing.
A breakthrough in asymmetric sulfurization came with the development of pentanidium-catalyzed reactions, which enable precise synthesis of sulfur-containing pharmacophores with high enantioselectivity [66]. This methodology addresses the long-standing challenge of synthesizing chiral sulfur-based compounds in single-enantiomer form, opening new possibilities for drug discovery programs seeking to exploit the versatile properties of sulfur-containing molecules without the risk of off-target effects from racemic mixtures.
Experimental Protocol: Sulfurization in Oligonucleotide Synthesis
Materials: Appropriate phosphoramidites, solid-phase synthesis support (CPG), acetonitrile (anhydrous), 0.25M 5-(ethylthio)-1H-tetrazole (ETT) activator solution, sulfurizing reagent (DDTT or Beaucage), deprotection reagents (aqueous methylamine, triethylammonium trihydrofluoride) [64].
Equipment: Automated DNA/RNA synthesizer (e.g., ABI 394), HPLC system with ion-exchange column (Dionex DNAPac PA200), ESI-mass spectrometer [64].
Procedure:
- Synthesizer Setup: Load sulfurizing reagent (0.05M DDTT or 0.05M Beaucage) to appropriate port on synthesizer. Use standard phosphoramidite coupling protocols with extended coupling time (4 minutes) when using TOM-protected or TBDMS-protected RNA phosphoramidites [64].
- Sulfurization Step: Program variable sulfurization times (30-240 seconds) to determine optimal conditions. For RNA synthesis, use 0.25M ETT as activator [64].
- Cleavage and Deprotection: After synthesis, cleave oligonucleotides from support using 1:1 mixture of 40% aqueous methylamine:33% ethanolic methylamine at room temperature overnight [64].
- Desilylation: For RNA oligonucleotides, dissolve dried oligos in dry DMSO, add triethylammonium trihydrofluoride, heat at 65°C for 1.5 hours [64].
- Purification and Analysis: Precipitate with butanol, wash with ethanol, and analyze by ion-exchange HPLC using sodium perchlorate gradient in TRIS buffer (pH 7.4) with 20% acetonitrile [64].
Performance Assessment: Calculate coupling efficiency based on HPLC purity and oligonucleotide length. Confirm complete sulfurization by ESI-mass spectrometry [64].
Diagram 2: Sulfurization workflow for oligonucleotide synthesis
Integrated Applications and Cross-Method Synergies
The strategic combination of mechanochemical activation with advanced sulfurization reagents creates powerful synergies for materials synthesis and pharmaceutical development. In one exemplary application, natural metal sulfide minerals like pyrite undergo mechanochemical activation to create highly efficient sorbents for vapor-phase mercury capture, achieving capacities exceeding 280 mg·g⁻¹ through the synergistic effect of structural defects and enhanced surface reactivity [62].
In pharmaceutical contexts, sulfur-containing pharmacophores generated through pentanidium-catalyzed asymmetric synthesis can be incorporated into drug discovery programs alongside mechanochemical activation to create novel therapeutic agents with improved metabolic stability and target specificity [66]. This integrated approach accelerates the discovery-to-development pipeline by enabling rapid generation of molecular diversity with controlled stereochemistry.
The convergence of these technologies is particularly impactful for sustainable chemistry initiatives, as both mechanochemical activation and modern sulfurization methods reduce or eliminate solvent requirements, minimize waste generation, and decrease energy consumption compared to traditional solution-based synthetic approaches.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Reagents for Mechanochemical and Sulfurization Studies
| Reagent/Material | Function/Application | Key Characteristics |
|---|---|---|
| Pentanidium Catalyst | Enantioselective sulfurization | Enables asymmetric synthesis of sulfur pharmacophores [66] |
| DDTT (Sulfurizing Reagent II) | Oligonucleotide phosphorothioate synthesis | 0.05M in acetonitrile, superior kinetics vs. Beaucage [64] |
| Sulfur Monochloride (S₂Cl₂) | Pharmaceutical sulfurization | Introduces sulfur groups into drug molecules [65] |
| Beaucage Reagent | Traditional sulfur transfer | 3H-1,2-benzodithiol-3-one-1,1-dioxide [64] |
| Elemental Sulfur (S₈) | Direct sulfurization agent | Used in thermal processes with hydrocarbons [67] |
| Copper Chloride (CuCl₂) | Mechanochemical additive | Enhances Hg capture capacity when ground with Fe₇S₈ [62] |
| Iron Powder | MCA co-additive | Increases copper extraction from chalcopyrite to 95.2% [61] |
Mechanochemical activation and advanced sulfurization reagents represent complementary pillars of modern solid-state synthesis, each offering distinct advantages for specific research and development applications. Mechanochemical approaches provide dramatic enhancements in reaction rates and material reactivity through microstructural modification and induced convective flows, while specialized sulfurization reagents enable precise incorporation of sulfur atoms with controlled stereochemistry.
The experimental data presented in this guide demonstrates that mechanochemical activation (MCA) consistently outperforms simple mechanical activation (MA) in enhancing reaction kinetics and product yields across multiple applications. Similarly, DDTT (Sulfurizing Reagent II) provides significant time savings and improved stability compared to traditional Beaucage Reagent for oligonucleotide synthesis. For asymmetric synthesis of pharmaceutical compounds, pentanidium-catalyzed sulfurization enables unprecedented control over chiral sulfur-containing pharmacophores.
Future developments in this field will likely focus on increasing the fundamental understanding of force-transduction mechanisms at molecular scales, developing increasingly selective sulfur-transfer reagents, and creating integrated platforms that combine mechanochemical processing with inline analysis and purification technologies. As these methodologies mature, their implementation across pharmaceutical development, materials science, and industrial chemistry will continue to expand, driven by their synergistic potential to address complex synthetic challenges while aligning with green chemistry principles.
Analytical Validation and Performance Benchmarking of Synthesized Materials
The development of advanced materials, particularly through solid-state synthesis routes, relies heavily on robust characterization techniques to understand microstructure, composition, and properties. As research into direct solid-state reaction flux methods advances, selecting appropriate characterization tools becomes paramount for elucidating reaction mechanisms, kinetics, and final product quality. This guide provides a comprehensive comparison of four cornerstone analytical techniques—X-ray Diffraction (XRD), Scanning Electron Microscopy (SEM), Impedance Spectroscopy, and Thermal Analysis—within the context of solid-state materials research. By objectively evaluating the capabilities, applications, and experimental requirements of each technique, this article aims to equip researchers and drug development professionals with the knowledge needed to select optimal characterization strategies for their specific investigative goals.
X-ray Diffraction (XRD)
X-ray Diffraction is a powerful nondestructive technique for characterizing crystalline materials based on the principle of constructive interference between X-rays and the periodic atomic arrangements within crystals. When X-rays impinge on a crystalline sample, they are scattered by the electrons of atoms. If the scattered waves satisfy Bragg's Law (nλ = 2d sinθ), where λ is the X-ray wavelength, d is the interplanar spacing, and θ is the diffraction angle, constructive interference occurs, producing detectable diffraction peaks [68] [69]. The resulting diffraction pattern serves as a unique fingerprint for crystalline phases present in the material, enabling phase identification, quantification, and structural analysis [70]. XRD is particularly valuable in solid-state reaction studies for tracking phase transformations, identifying intermediate compounds, and determining reaction completeness.
Scanning Electron Microscopy (SEM)
Scanning Electron Microscopy provides high-resolution surface imaging by scanning a focused electron beam across a specimen and detecting various signals generated by electron-matter interactions [71]. Unlike optical microscopes limited by light wavelength, SEM uses electrons with much shorter wavelengths, enabling resolution down to the nanometer scale [71]. Key signals include secondary electrons for topographical contrast, backscattered electrons for compositional contrast based on atomic number differences, and characteristic X-rays for elemental analysis via Energy Dispersive X-ray Spectroscopy (EDS) [72] [71]. For solid-state reaction studies, SEM reveals morphological evolution, particle size distribution, and surface characteristics, while EDS provides elemental mapping to assess reaction homogeneity and diffusion profiles.
Impedance Spectroscopy
Electrochemical Impedance Spectroscopy (EIS) characterizes the electrical response of materials by applying a small amplitude AC potential and measuring the current response across a frequency range [73]. Unlike simple resistance measurement, impedance accounts for frequency-dependent behavior, representing data as complex quantities (Z = Z' + jZ") that can be correlated with underlying electrochemical processes [73]. EIS data is typically presented as Nyquist plots (imaginary vs. real components) or Bode plots (impedance magnitude and phase shift vs. frequency), and analyzed using equivalent circuit models comprising resistors, capacitors, and inductors that represent physical processes within the material [73]. For solid-state ionics and materials research, EIS is indispensable for determining conductivity, characterizing interfacial phenomena, and investigating charge transport mechanisms.
Thermal Analysis
Thermal Analysis encompasses a suite of techniques that measure physical and chemical property changes as functions of temperature. Key methods include Differential Scanning Calorimetry (DSC) measuring heat flows associated with transitions; Thermogravimetric Analysis (TGA) monitoring mass changes; Thermomechanical Analysis (TMA) assessing dimensional changes; and Dynamic Mechanical Analysis (DMA) determining viscoelastic properties under oscillatory stress [74]. These techniques reveal thermal stability, decomposition behavior, phase transitions, glass transitions, and compositional information, making them vital for optimizing solid-state synthesis conditions and understanding material behavior under thermal processing.
Technical Comparison and Experimental Data
The following tables provide a detailed technical comparison of the four characterization techniques, highlighting their specific applications in solid-state reaction flux research.
Table 1: Core Capabilities and Applications in Solid-State Reaction Studies
| Technique | Primary Measured Parameters | Solid-State Reaction Applications | Key Limitations |
|---|---|---|---|
| XRD | Peak position, intensity, width, and shape [68] [70] | Phase identification and quantification [70], crystal structure determination [69], tracking phase transformations [75], crystallite size calculation [70] | Limited to crystalline materials; weak for amorphous phases; surface-insensitive |
| SEM/EDS | Secondary electron yield, backscattered electron coefficient, X-ray energy [72] [71] | Morphology analysis [75], particle size distribution, elemental composition and mapping [72], reaction interface examination, homogeneity assessment | Requires conductive coatings for insulating samples; high vacuum typically needed; sample size restrictions |
| Impedance Spectroscopy | Complex impedance (Z' and Z"), phase angle, modulus [73] | Ionic/electronic conductivity measurement [73], defect characterization, grain boundary contributions, interfacial reaction kinetics | Requires electrode contacts; complex data interpretation; limited to electrically active materials |
| Thermal Analysis | Heat flow (DSC), mass change (TGA), dimensional change (TMA), modulus (DMA) [74] | Reaction kinetics studies, thermal stability assessment [74], decomposition temperature determination, glass transition detection [74] | Small sample sizes; potential kinetic effects from heating rates; complementary techniques often needed |
Table 2: Experimental Requirements and Typical Operating Parameters
| Technique | Sample Preparation Requirements | Typical Experimental Conditions | Output Data Format |
|---|---|---|---|
| XRD | Powder (<10µm) or flat solid surface; minimal preparation [69] | Ambient conditions; Cu Kα radiation (λ=1.54Å); 5-80° 2θ range [75] | Diffractogram (Intensity vs. 2θ) [68] |
| SEM/EDS | Conductive coating for non-conductive samples; specific size (cm-scale) [72] | High vacuum (10⁻³ to 10⁻⁶ Torr); accelerating voltage 1-30 kV [71] | Grayscale images; elemental spectra/maps |
| Impedance Spectroscopy | Electrode application (sputtering, painting); controlled geometry [73] | Frequency range 0.01 Hz-1 MHz; AC amplitude 1-10 mV; variable temperature [73] | Complex impedance plots (Nyquist, Bode) |
| Thermal Analysis | Specific mass (mg-range) or geometry; contact with sensor [74] | Temperature range -150°C to 1600°C; heating rates 1-20°C/min; various atmospheres [74] | Thermograms (Property vs. Temperature) |
Table 3: Quantitative Data from Representative Solid-State Reaction Studies
| Technique | Material System | Key Measured Values | Experimental Findings |
|---|---|---|---|
| XRD | Ye'elimite (3CaO·3Al₂O₃·CaSO₄) hydration products [75] | d-spacings, phase identification | Identified ettringite, monosulfoaluminate, and gibbsite as long-term hydration products after 270 days [75] |
| SEM/EDS | Ye'elimite hydration at w/y ratio of 3 [75] | Morphological features, elemental ratios | Revealed co-precipitation of hydration products and heterogeneous growth mechanisms; EDS confirmed chemical composition [75] |
| TGA/DSC | Polymer composites [74] | Decomposition temperature: 300-500°C; glass transition: -50 to 150°C [74] | Determined thermal stability, filler content, and phase transition temperatures for material selection [74] |
| Impedance Spectroscopy | Solid electrolytes [73] | Bulk resistance: 10-10⁶ Ω; capacitance: 10⁻¹²-10⁻⁶ F [73] | Separated bulk, grain boundary, and electrode contributions to total resistance using equivalent circuit modeling [73] |
Experimental Protocols and Methodologies
XRD Sample Preparation and Analysis Protocol
For solid-state reaction studies, proper sample preparation is crucial for obtaining meaningful XRD results. The standard methodology involves:
Sample Preparation: Grind the solid-state reaction product to a fine powder (typically <10 micrometers) using an agate mortar and pestle to minimize preferred orientation effects and ensure adequate particle statistics [69].
Mounting: Pack the powder into a sample holder using a back-loading technique to create a flat, level surface with random crystallite orientation.
Data Collection: Operate the diffractometer with Cu Kα radiation (λ = 1.5418 Å) at 40 kV and 40 mA. Scan typically from 5° to 80° 2θ with a step size of 0.02° and a counting time of 1-2 seconds per step [75].
Phase Identification: Compare collected diffraction patterns with reference patterns in the International Centre for Diffraction Data (ICDD) database using search-match software [70] [69]. Identify all major and minor phases present in the reaction product.
Quantitative Analysis: Employ Rietveld refinement methods for accurate phase quantification, particularly important for tracking reaction progress in solid-state synthesis.
Combined SEM-EDS Analysis for Solid-State Reaction Products
The integration of SEM with EDS provides comprehensive microstructural and compositional information:
Sample Preparation: Mount solid samples on aluminum stubs using conductive carbon tape. For non-conductive materials, apply a thin (5-10 nm) coating of gold or carbon using sputter coating to prevent charging effects [72] [71].
Imaging Conditions: Operate the SEM at an accelerating voltage of 10-20 kV for optimal resolution and adequate X-ray generation. Use both secondary electron (SE) and backscattered electron (BSE) detectors to obtain topographic and compositional contrast, respectively [71].
Elemental Analysis: For EDS, collect spectra from multiple regions of interest with sufficient counting time to ensure good statistics. Perform elemental mapping to visualize distribution of specific elements across the sample surface, particularly useful for identifying reaction zones and diffusion profiles in solid-state reactions [75] [72].
Data Interpretation: Correlate morphological features observed in SEM images with compositional data from EDS to understand reaction mechanisms and heterogeneity in solid-state synthesis products.
Impedance Spectroscopy Measurement Methodology
For accurate impedance characterization of solid-state materials:
Electrode Application: Apply symmetric electrodes to opposite faces of pelletized samples using sputtered gold, painted silver paste, or spring-loaded contacts, depending on material properties and measurement temperature [73].
Experimental Setup: Connect the sample to an impedance analyzer using a two-probe or four-probe configuration. Place the assembly in a temperature-controlled furnace for elevated temperature measurements.
Data Acquisition: Apply a small AC signal (typically 10-1000 mV) across a frequency range from 0.01 Hz to 1 MHz, ensuring the measurement is within the linear response regime of the material [73]. Collect data at temperature intervals of 25-50°C to determine activation energies for conduction.
Equivalent Circuit Modeling: Fit the impedance data to appropriate equivalent circuits using specialized software. Common circuit elements include resistors (R) for conduction processes, constant phase elements (CPE) for non-ideal capacitance, and Warburg elements (W) for diffusion-controlled processes [73].
Thermal Analysis Protocol for Solid-State Reaction Kinetics
Thermal analysis techniques provide critical information about reaction behavior:
Sample Preparation: For TGA, use 10-20 mg of finely powdered sample in an open alumina crucible. For DSC, use hermetically sealed pans with 5-10 mg sample amounts for accurate heat flow measurement [74].
Temperature Program: Employ heating rates of 5-20°C/min under controlled atmosphere (nitrogen for inert conditions, air for oxidative environments). For reaction kinetics studies, multiple heating rates are recommended for Kissinger or Ozawa analysis [74].
Data Interpretation: Identify key thermal events: endothermic peaks (melting, decomposition) and exothermic peaks (crystallization, oxidation) in DSC; mass loss steps (dehydration, decomposition) in TGA. Correlate events between techniques for comprehensive understanding.
Kinetic Analysis: For solid-state reaction studies, apply model-fitting or model-free methods to determine activation energies and reaction mechanisms from thermal analysis data.
Experimental Workflows and Technical Pathways
The following diagrams illustrate standard experimental workflows and decision processes for materials characterization in solid-state reaction studies.
XRD Analysis Workflow
SEM-EDS Analysis Workflow
Multi-Technique Characterization Approach
Research Reagent Solutions and Essential Materials
Table 4: Essential Materials and Reagents for Materials Characterization
| Item | Function/Application | Technical Specifications |
|---|---|---|
| XRD Sample Holders | Secure powder samples during diffraction analysis | Aluminum or glass holders with cavity depth of 0.2-1.0 mm; zero-background plates for preferred orientation minimization |
| Conductive Coatings | Render non-conductive samples suitable for SEM imaging | Gold/palladium (5-20 nm thickness) for high-resolution imaging; carbon (10-30 nm) for EDS analysis to avoid interference |
| SEM Mounting Supplies | Secure samples to SEM stubs | Aluminum stubs (various diameters); conductive carbon tape; silver paint for improved electrical contact |
| Impedance Electrode Materials | Create electrical contacts for impedance measurements | Sputtered gold electrodes (50-100 nm) for blocking contacts; silver paste for porous electrodes; spring-loaded contacts for high-temperature measurements |
| Thermal Analysis Crucibles | Contain samples during thermal measurements | Aluminum pans (DSC, hermetic and non-hermetic); alumina crucibles (TGA, up to 1600°C); platinum crucibles for corrosive samples |
| Standard Reference Materials | Instrument calibration and method validation | Silicon powder (XRD angle calibration); alumina (thermal conductivity standards); pure metals (DSC temperature calibration) |
| Polishing Supplies | Prepare cross-sections for microstructural analysis | Silicon carbide papers (120-4000 grit); diamond suspensions (1-0.25 µm); colloidal silica for final polishing |
The comprehensive comparison presented in this guide demonstrates that XRD, SEM, Impedance Spectroscopy, and Thermal Analysis offer complementary capabilities for characterizing solid-state reaction products. XRD provides essential phase identification and structural information, SEM reveals critical microstructural and compositional details, Impedance Spectroscopy elucidates electrical properties and transport mechanisms, while Thermal Analysis delivers vital information on stability and reaction behavior. The optimal characterization strategy employs an integrated approach, leveraging the strengths of each technique to develop a complete understanding of material properties and reaction mechanisms. As solid-state synthesis methods continue to evolve, particularly in pharmaceutical development and advanced materials research, these characterization techniques will remain fundamental tools for elucidating structure-property relationships and optimizing synthesis protocols.
Comparative Analysis of Photocatalytic H2 Evolution Performance
The efficient production of hydrogen via photocatalytic water splitting represents a cornerstone of sustainable energy research. Central to advancing this technology is the development of high-performance photocatalysts and the synthesis methods used to produce them. The choice of synthesis technique profoundly influences critical material properties, including crystallinity, particle size, defect density, and ultimately, photocatalytic hydrogen evolution performance. This review provides a comparative analysis of various photocatalytic materials, with a particular focus on the impact of synthesis methodologies, especially the contrast between direct solid-state reactions and flux-assisted methods. By examining quantitative performance data and detailed experimental protocols, this guide aims to inform researchers and development professionals in selecting and optimizing synthesis routes for enhanced photocatalytic activity.
Performance Comparison of Photocatalytic Materials
The hydrogen evolution performance of various photocatalysts varies significantly based on their composition, structure, and synthesis method. The following table summarizes the performance of several prominent catalysts as reported in recent literature.
Table 1: Comparative Hydrogen Evolution Performance of Selected Photocatalysts
| Photocatalyst | Co-catalyst / Modification | Synthesis Method | H2 Evolution Rate | Light Source & Conditions | Reference |
|---|---|---|---|---|---|
| Y₂Ti₂O₅S₂ (YTOS) | Not specified | CS₂ sulfurization with CaCl₂ flux | Higher than SSR and H₂S methods | Simulated solar light, sacrificial donor | [33] |
| Pt-SA/NP-TiO₂-xBₓ | B-Pt-O asymmetric coordination | Thermal treatment of MOF precursor | 627.6 mmol g⁻¹ h⁻¹ | Apparent Quantum Efficiency: 98.4% @ 365 nm | [76] |
| Mg/Fe-LDH | None | Co-precipitation | 2542.36 mmol h⁻¹ cm⁻² | 460 nm & 490 nm illumination | [77] |
| Zn₀.₅Cd₀.₅S/CoWO₄ (10%) | S-type heterojunction | Solvothermal | 223.30 μmol g⁻¹ h⁻¹ (with PP-ol); 140.28 μmol g⁻¹ h⁻¹ (with sodium lignosulfonate) | Visible light, biomass reforming | [78] |
| MNb₂O₆-based composites (e.g., with g-C₃N₄ or TiO₂) | Heterostructure engineering | Hydrothermal, solvothermal, electrospinning | Up to 146 mmol h⁻¹ g⁻¹ | Visible light | [79] |
The data reveals several important trends. Pt-based catalysts, particularly those with sophisticated atomic-scale engineering like asymmetric coordination, achieve exceptionally high hydrogen evolution rates and nearly unity apparent quantum efficiency [76]. Layered Double Hydroxides (LDHs), such as Mg/Fe-LDH, also demonstrate remarkable performance per unit area, attributed to their high surface area and abundant active sites [77]. In contrast, metal sulfide and oxysulfide systems like YTOS and ZnCdS/CoWO4, while exhibiting more modest absolute rates, remain crucial for applications utilizing visible light and for synergistic processes like biomass reforming [33] [78]. The performance of niobate-based materials (MNb₂O₆) can be significantly boosted through heterostructure formation [79].
The Synthesis Method Paradigm: Solid-State vs. Flux-Assisted Routes
The synthesis methodology is a critical determinant of a photocatalyst's physical and electronic properties. The conventional Solid-State Reaction (SSR) method involves thoroughly mixing solid precursors (e.g., Y₂O₃, Y₂S₃, and TiO₂ for YTOS) and calcining them at high temperatures in sealed, evacuated quartz tubes for extended durations, often exceeding four days [33]. This method suffers from low mass transfer rates, often resulting in large particle sizes (micrometers to tens of micrometers) and a high density of bulk defects that act as charge recombination centers [33].
The Flux-Assisted Method was developed to address these limitations. This technique involves mixing the solid precursors with a molten salt flux (e.g., CaCl₂, CsCl, LiCl/MgCl₂) before calcination. The ionic molten salt acts as a polar solvent that enhances mass transfer by dissolving reactant species, facilitating their rapid migration via convection and diffusion [33]. The key advantages of this method include:
- Reduced Particle Size: Significantly decreases particle dimensions, shortening the migration distance of photogenerated charges to the surface [33].
- Enhanced Crystallinity: Promotes the growth of high-quality crystals with fewer bulk defects, reducing charge recombination [33] [79].
- Shorter Reaction Times: The enhanced mass transfer can reduce calcination times from several days to just hours [33].
A recent advancement is the CS₂ Sulfurization Method combined with a flux. This approach uses gaseous carbon disulfide (CS₂) as a sulfurizing agent, which offers high sulfurization activity. When applied to oxide precursors in the presence of a flux like CaCl₂, it enables the formation of oxysulfides like YTOS at lower temperatures (e.g., 800°C) compared to H₂S sulfurization [33]. This method combines the benefits of gas-solid reactions (improved kinetics and smaller particles) with the crystallinity-enhancing effects of a flux, resulting in materials with superior photocatalytic activity compared to those from SSR or H₂S methods [33]. The workflow below illustrates the progression from traditional to advanced synthesis methods.
Detailed Experimental Protocols
Flux-Assisted CS₂ Sulfurization of Y₂Ti₂O₅S₂ (YTOS)
The synthesis of high-performance YTOS via flux-assisted CS₂ sulfurization involves several critical stages [33]:
- Precursor Preparation: The starting materials can be a mixture of Y₂O₃, Y₂S₃, and TiO₂ in a specific molar ratio (e.g., 1:2:6), or simply a mixture of Y₂O₃ and TiO₂ (1:2 molar ratio), or pre-synthesized Y₂Ti₂O₇.
- Flux Integration: The precursor mixture is thoroughly ground with a flux agent (e.g., 500 wt% CaCl₂) in an inert atmosphere glovebox for 30 minutes.
- Sulfurization Reaction: The mixture is transferred to an alumina boat and placed in a horizontal tube furnace. A flow of N₂ gas is passed through a CS₂ reservoir (held at 10°C for stable evaporation) to create a CS₂/N₂ gas mixture, which is then directed into the furnace. The temperature is ramped (e.g., 10°C/min to 500°C, then 5°C/min to 800°C) and held for several hours (e.g., 3 hours).
- Post-processing: After cooling, the CS₂ flow is stopped and the sample is purged with N₂. The resulting powder is sonicated in distilled water, and the flux is removed by filtration. The final product is dried.
Synthesis of S-scheme ZnCdS/CoWO₄ Heterojunction
The construction of this heterojunction photocatalyst is a multi-step process [78]:
- Synthesis of Zn₀.₅Cd₀.₅S (ZCS) Nanoparticles: Zn(CH₃COO)₂·2H₂O, Cd(CH₃COO)₂·2H₂O, and thioacetamide (C₂H₅NS) are dissolved in deionized water. NaOH solution is added dropwise under constant stirring. The suspension is transferred to a Teflon-lined autoclave and maintained at 180°C for 24 hours. The resulting yellow precipitate is washed and dried.
- Synthesis of CoWO₄ (CW) Nanocrystals: Co(NO₃)₂·6H₂O and Na₂WO₄·2H₂O are dissolved in deionized water and stirred to form a homogeneous dispersion. The mixture is heated in an autoclave at 180°C for 24 hours. The blue product is washed and dried.
- Construction of ZCS/CW Heterojunction: The as-prepared ZCS and CW powders are mixed in a mass ratio of 10:1 in ethanol. The mixture is stirred and heated in a water bath at 80°C to evaporate the solvent. The final composite is dried at 60°C.
The Scientist's Toolkit: Essential Research Reagents
The following table lists key reagents and materials commonly used in the synthesis and testing of advanced photocatalysts, as evidenced by the reviewed protocols.
Table 2: Key Research Reagents for Photocatalyst Synthesis and Testing
| Reagent/Material | Function in Research | Example Use Case |
|---|---|---|
| Carbon Disulfide (CS₂) | Highly active sulfurizing reagent | Low-temperature synthesis of Y₂Ti₂O₅S₂ oxysulfide [33] |
| Molten Salt Fluxes (e.g., CaCl₂, CsCl) | Enhances mass transfer, reduces synthesis temperature & time, improves crystallinity | Flux-assisted synthesis of YTOS and Sm₂Ti₂O₅S₂ [33] |
| Metal-Organic Frameworks (MOFs) | Precursors for creating tailored structures with high surface area and dispersed active sites | Synthesis of PtSA/NP-TiO₂-xBₓ with asymmetric coordination [76] |
| Sacrificial Reagents (e.g., Na₂SO₃, lactic acid) | Electron donors that consume photogenerated holes, enhancing proton reduction efficiency | Standard component in photocatalytic H₂ evolution testing setups [78] [77] |
| Noble Metal Co-catalysts (e.g., Pt, Au) | Facilitate charge separation and lower overpotential for H₂ evolution | Pt nanoparticles and single atoms in TiO₂-based systems [76] [80] |
Charge Transfer Mechanisms in Heterostructures
A key strategy for improving photocatalytic performance is engineering heterojunctions to manage charge carrier separation. The following diagram illustrates two common charge transfer pathways.
The mechanistic pathways include:
- Type-II Heterojunction: Electrons migrate from the higher conduction band (CBM) of one semiconductor (SC-I) to the lower CBM of another (SC-II), while holes move in the opposite direction from the valence band (VBM of SC-II to VBM of SC-I). This spatially separates charge carriers but can compromise the redox potential of the system [81].
- S-Scheme (Step-Scheme) Heterojunction: This advanced configuration preserves the most useful charge carriers with the strongest redox power. The internal electric field at the interface promotes the recombination of less useful electrons and holes, leaving the strongest reductants (in the reduction photocatalyst's CBM) and oxidants (in the oxidation photocatalyst's VBM) to participate in reactions [78] [80]. This mechanism is often cited for systems like ZnCdS/CoWO₄ [78].
This comparative analysis underscores that the photocatalytic hydrogen evolution performance is intrinsically linked to the synthesis methodology and architectural design of the material. While traditional solid-state reactions often produce materials with suboptimal properties, advanced techniques like flux-assisted and gas-phase sulfurization methods yield catalysts with superior crystallinity, optimal particle size, and consequently, enhanced activity. The ongoing refinement of synthesis protocols, coupled with sophisticated heterojunction engineering and atomic-scale modulation of active sites, continues to push the boundaries of photocatalytic efficiency. Future research should focus on scaling these advanced synthesis methods, reducing reliance on scarce elements, and further elucidating reaction mechanisms to enable the large-scale deployment of photocatalytic hydrogen production technologies.
Benchmarking Electrophysical Properties for Energy Storage
The pursuit of advanced energy storage solutions is intrinsically linked to the development of superior battery materials. The performance of these materials—their capacity, rate capability, and cycle life—is governed by their fundamental electrophysical properties, which are in turn dictated by their synthesis route. Within this context, direct solid-state reaction methods represent a foundational class of synthesis techniques for producing polycrystalline cathode materials [1]. These methods involve reactions between solid precursors at high temperatures and are prized for their simplicity and scalability for industrial production [1].
This guide provides a comparative analysis of electrode materials synthesized via solid-state reactions, benchmarking their key electrophysical properties against alternatives. It is structured to serve researchers and scientists by presenting clearly organized quantitative data, detailed experimental protocols, and essential resource information to inform material selection and research design in drug development and related fields.
Synthesis Workflow: Solid-State Reaction Method
The following diagram illustrates the generalized experimental workflow for synthesizing cathode materials via a solid-state reaction, as exemplified by the production of lithium nickel manganese oxide (LNMO) hollow microspheres [1].
Diagram Title: Solid-State Synthesis Workflow
This process leverages phenomena like the Kirkendall effect, where the differential diffusion rates of metal ions (e.g., Mn and Ni) and oxygen atoms lead to the formation of hollow microstructures, a key morphological feature that enhances electrochemical performance [1].
Comparative Performance of Energy Storage Materials
The electrophysical performance of energy storage materials is quantified through key metrics. The table below summarizes the performance of selected materials synthesized via solid-state and other methods, based on experimental data from the literature.
Table 1: Performance Benchmarking of Energy Storage Materials
| Material & Synthesis Method | Specific Capacity (mAh/g) | Cycle Life (Cycles) | Capacity Retention | Key Electrophysical Properties | Ref |
|---|---|---|---|---|---|
| LNMO Hollow Microspheres (Solid-State) | 118 (at 1C) | 200 | 96.6% (at 2C) | Short Li+ diffusion path, porous structure | [1] |
| LFP/C Composite (Solid-State) | 167.3 (at 0.1C) | 100 | Good retention reported | Enhanced conductivity from graphitic carbon | [1] |
| Ag-doped NiCo₂O₄ (Hydrothermal) | 1501 F/g (Capacitance) | 5000 | 106% | Low solution & charge transfer resistance | [82] |
| CS/PET Composite (Hydrothermal) | 193 F/g (Capacitance) | 2500 | 92% | Sustainable, oxygen-rich carbon composite | [82] |
| PVA Hydrogel Zn-Air | N/A | 34h (stable) | N/A | High round-trip efficiency, flexible | [82] |
Performance data reveals that materials produced via solid-state reactions, particularly those with engineered hollow or porous morphologies, achieve excellent capacity retention due to their short lithium-ion diffusion paths and robust structures that accommodate volume changes during cycling [1].
Performance Metrics for System-Level Comparison
When evaluating complete energy storage systems, performance is measured by a different set of Key Performance Indicators (KPIs) that relate to overall efficiency, lifespan, and practicality.
Table 2: Key Performance Indicators for Energy Storage Systems
| KPI | Definition & Measurement | Impact on Performance & Value | |
|---|---|---|---|
| Cycle Life | Number of charge-discharge cycles before capacity drops to 80% of original. | Directly determines system lifespan and levelized cost of storage (LCOS). Systems can exceed 6,000 cycles. | [83] |
| Depth of Discharge (DoD) | Percentage of total capacity used per cycle (e.g., 80% DoD). | Deeper discharge increases usable energy but can stress the battery, impacting cycle life. | [84] [83] |
| Round-Trip Efficiency (RTE) | Ratio of energy output to input over a full cycle (e.g., 92%). | Higher RTE means less energy wasted as heat, leading to greater operational cost savings. | [83] [82] |
| C-Rate | Rate of charge/discharge relative to capacity (e.g., 1C, 0.5C). | Determines application fit: low C-rates (0.25C) for energy storage, high C-rates (1C+) for power applications. | [84] [83] |
These KPIs are interconnected; for instance, operating a system at a very high DoD may reduce its cycle life, while a low RTE increases the effective cost of stored energy [83].
The Researcher's Toolkit: Essential Reagents & Materials
Successful synthesis and testing of energy storage materials rely on a set of core reagents and instruments.
Table 3: Key Research Reagent Solutions and Materials
| Item | Function in Research Context | Example Application | |
|---|---|---|---|
| Lithium Hydroxide (LiOH) | Lithium precursor in solid-state synthesis of cathode materials. | Synthesis of LNMO and LFP cathode materials. | [1] |
| Transition Metal Salts | Source of transition metal ions (e.g., Ni, Mn, Co, Fe) in the crystal structure. | Ni(NO₃)₂ and MnO₂ used as precursors for LNMO synthesis. | [1] |
| Carbonaceous Surfactants | Act as carbon sources in-situ during pyrolysis, forming conductive networks. | Tween series surfactants used to create LFP/C composites. | [1] |
| Hydrogel Substrates | Serve as integrated electrolyte and electrode matrices in flexible batteries. | PVA hydrogel used in flexible zinc-air batteries. | [82] |
| Phase Change Materials | Store thermal energy via latent heat, useful for thermal management of batteries. | Erythritol-based composites for medium-temperature storage. | [82] |
The benchmarking data and methodologies presented herein underscore a critical principle in energy storage research: the synthesis pathway, such as the solid-state reaction method, is a primary determinant of a material's ultimate electrophysical characteristics. The ability of solid-state reactions to produce materials with tailored morphologies—such as the hollow LNMO microspheres—confers significant performance advantages in key areas like cycle life and rate capability. For researchers, the strategic selection of a synthesis method, guided by a clear understanding of the target KPIs and supported by the appropriate reagents and experimental protocols, is paramount to developing next-generation energy storage solutions that meet the demanding requirements of modern applications.
Evaluating Crystallinity, Defect Density, and Ionic Conductivity
Within the field of materials science, particularly for energy storage and pharmaceutical applications, the performance of solid-state materials is critically dependent on key structural and transport properties. This guide provides a comparative analysis of the methodologies for evaluating three fundamental parameters—crystallinity, defect density, and ionic conductivity—in the context of synthesizing materials via direct solid-state reaction flux methods. Solid-state metathesis reactions have emerged as a powerful synthetic route, enabling the production of metastable materials and polymorphs, such as Mg- and Ca-based ion conductors, at significantly lower temperatures and pressures than traditional high-temperature solid-state reactions [85]. Objectively comparing the performance of materials synthesized through these alternative pathways requires a unified understanding of their structural integrity, compositional purity, and functional efficacy. This guide details the experimental protocols and data interpretation necessary for such comparisons, providing a framework for researchers and development professionals to validate and select superior synthetic routes.
Experimental Protocols for Key Characterization Techniques
Protocol for Crystallinity Analysis via X-ray Diffraction (XRD)
Principle: X-ray Diffraction (XRD) quantifies the degree of crystallinity by differentiating the sharp diffraction peaks from crystalline regions from the broad scattering halos from amorphous regions [86].
Procedure:
- Sample Preparation: Prepare a flat, homogeneous powder sample of the material to be analyzed.
- Data Acquisition: Irradiate the sample with a collimated X-ray beam and record the diffraction pattern across a relevant 2θ range (e.g., 5° to 60°).
- Pattern Deconvolution: Fit the resulting XRD pattern to separate the integrated intensity of all crystalline peaks ((Ic)) from the integrated intensity of the amorphous halo ((Ia)).
- Calculation: The degree of crystallinity ((Xc)) is calculated using the formula: (Xc (\%) = \frac{Ic}{(Ic + I_a)} \times 100) [86].
Alternative Protocol: Crystallinity by Density Measurement This method requires knowledge of the densities of perfectly crystalline ((\rhoc)) and perfectly amorphous ((\rhoa)) samples of the same material.
- Density Measurement: Precisely measure the density ((\rho)) of your sample using a technique like Archimedes' principle [87].
- Calculation: The weight fraction crystallinity ((\chi)) is calculated as: (\chi = \frac{\rho - \rhoa}{\rhoc - \rhoa} \times \frac{\rhoc}{\rho}) [87].
Protocol for Defect Density Analysis via Transmission Electron Microscopy (TEM)
Principle: Defect density, such as the number of dislocations or vacancies per unit volume, can be quantified using diffraction contrast techniques in TEM, which are sensitive to lattice strain around defects [88].
Procedure:
- Sample Preparation: Prepare electron-transparent thin foils of the material using focused ion beam (FIB) milling or electropolishing.
- Imaging Mode Selection:
- Two-Beam Bright-Field (BF): Tilt the sample to a strong two-beam condition to image dislocations with good contrast [88].
- Weak-Beam Dark-Field (WBDF): Use a high-order diffraction vector for higher resolution imaging of fine defects [88].
- Down-Zone STEM: Image with the beam aligned along a low-index zone axis to visualize defect structures across many grains simultaneously [88].
- Defect Identification and Counting: Acquire multiple images from different areas and under different diffraction vectors. Manually or automatically count the number of defects (e.g., dislocation lines) in each image.
- Calculation: The defect density ((\rhod)) for line defects is given by (\rhod = \frac{2N}{Lt}), where (N) is the number of intersections between dislocations and random test lines, (L) is the total length of test lines, and (t) is the sample thickness [88].
Protocol for Ionic Conductivity Measurement via Electrochemical Impedance Spectroscopy (EIS)
Principle: Ionic conductivity ((\sigma)) measures a material's ability to conduct ions and is determined by applying a small AC voltage across a sample and analyzing the resistance.
Procedure:
- Cell Fabrication: Synthesize the material into a dense pellet. Apply ion-blocking electrodes (e.g., gold, platinum) to both faces of the pellet to form a symmetric cell.
- Impedance Measurement: Place the cell in a temperature-controlled fixture. Use an impedance analyzer to measure the electrochemical impedance spectrum over a wide frequency range (e.g., 1 MHz to 0.1 Hz) at a specified voltage amplitude.
- Data Fitting: Fit the resulting Nyquist plot (typically featuring a semicircle and a spike) with an equivalent circuit model (e.g., a resistor in parallel with a constant phase element, followed by a Warburg element).
- Calculation: Extract the bulk resistance ((Rb)) from the high-frequency intercept of the semicircle with the real axis. The ionic conductivity is calculated as (\sigma = \frac{l}{Rb \times A}), where (l) is the pellet thickness and (A) is the electrode area [89] [85].
Comparative Performance Data
The following tables consolidate typical data and performance indicators for the characterized properties across different classes of materials, providing a benchmark for comparison.
Table 1: Crystallinity and Ionic Conductivity Data for Selected Solid Electrolytes
| Material Class / Example | Synthesis Method | Crystallinity (%) | Ionic Conductivity (S cm⁻¹) | Measurement Temperature | Citation |
|---|---|---|---|---|---|
| Organic-Inorganic Composite (PEO:LLZTO) | Solution Casting | N/A | (2.1 \times 10^{-4}) | 30 °C | [89] |
| Inorganic (Ilmenite-type Mg₃TeO₆) | Solid-State Metathesis | Assumed High | Remarkable bulk ionic conductivity (Mg²⁺) | Room Temperature | [85] |
| Inorganic (Double Perovskite Ca₂MgTeO₆) | Solid-State Metathesis | Assumed High | Remarkable bulk ionic conductivity (Ca²⁺) | Room Temperature | [85] |
Table 2: Defect Density Ranges in Various Materials
| Material | Defect Type | Defect Density | Measurement Technique | Context | Citation |
|---|---|---|---|---|---|
| Nanocrystalline Au | Irradiation-induced defects | Quantified | ACOM-assisted TEM, WBDF-TEM | After irradiation | [88] |
| Android Kernel Code | Software bugs | ~0.47 defects/KLOC | Static Analysis (Coverity) | Industry benchmark | [90] |
| Sb₂Se₃ thin films | Shallow acceptor SeSb | ~10¹⁶ cm⁻¹eV⁻¹ at peak | Thermal Admittance Spectroscopy | As-fabricated | [90] |
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagents and Materials for Synthesis and Characterization
| Item | Function/Brief Explanation | Typical Application |
|---|---|---|
| Honeycomb Layered Oxides (e.g., Li₄MgTeO₆, Na₂Mg₂TeO₆) | Precursors for solid-state metathesis reactions to form novel Mg²⁺/Ca²⁺ conductors [85]. | Synthesis |
| Alkaline-Earth Salts (e.g., MgCl₂, Mg(NO₃)₂, Ca(NO₃)₂) | Reactants in metathesis reactions, enabling low-temperature synthesis pathways [85]. | Synthesis |
| Inorganic Fillers (e.g., Al₂O₃, LLZO (Li₇La₃Zr₂O₁₂)) | Enhance ionic conductivity, mechanical strength, and interfacial stability in composite solid electrolytes [89]. | Composite Fabrication |
| Ion-Blocking Electrodes (e.g., Gold, Platinum) | Serve as inert contacts for EIS measurements, blocking ion passage and forcing ionic current through the bulk. | Ionic Conductivity (EIS) |
| XRD Standard Reference Materials | Used for instrument calibration to ensure accurate peak position and intensity data. | Crystallinity (XRD) |
| TEM Calibration Specimens (e.g., Au nanoparticles on carbon) | Used to calibrate magnification and camera constant of the TEM. | Defect Density (TEM) |
Experimental Workflow and Relationship Diagram
The following diagram illustrates the logical sequence and relationships between the synthesis method, the key characterization techniques discussed, and the final material properties evaluated.
Characterization Workflow
This workflow outlines the standard process for evaluating materials, beginning with synthesis and moving through parallel characterization of key properties to inform final performance assessment.
Ionic Conduction Mechanism in Composite Electrolytes
The addition of inorganic fillers to polymer matrices, forming organic-inorganic composite solid electrolytes (OICSEs), enhances ionic conductivity through multiple mechanisms. The following diagram delineates these functional mechanisms.
Filler Enhancement Mechanisms
This diagram details how inorganic fillers like active LLZO or inert Al₂O3 enhance OICSE performance. Key mechanisms include disrupting polymer crystallinity to create more amorphous pathways for ion transport [89], engaging in Lewis acid-base interactions to dissociate lithium salts and increase Li⁺ transference number [89], and for active fillers, creating percolation networks for rapid ion conduction [89]. Additionally, fillers improve mechanical strength to suppress lithium dendrite growth [89].
The objective comparison of material performance in scientific literature is often complicated by inconsistent synthesis methodologies. Small variations in solid-state synthesis parameters can lead to significant differences in a material's fundamental characteristics, thereby obscuring direct performance comparisons. This guide establishes a framework for accounting for these critical synthesis conditions, enabling fair and meaningful comparisons between materials, with a specific focus on direct solid-state reaction methods.
The solid-state reaction route is a cornerstone of materials synthesis, valued for its simplicity and large-scale production capabilities [1]. However, this method presents inherent challenges for comparative studies, including poor control over final size and shape, difficulty obtaining nanostructured materials with well-controlled morphology, and potential inhomogeneity due to imperfect mixing of solid reagents [1]. Without proper standardization, these factors introduce variability that compromises the validity of performance comparisons.
Fundamental Principles of Solid-State Synthesis
Core Mechanism and Influencing Factors
Solid-state synthesis involves a chemical reaction between solid precursors to form a new solid-phase material, typically initiated and sustained at elevated temperatures. The success of this process depends on diffusional exchange among grains of the reagents, which leads to structural changes in the material [91].
Several critical factors influence solid-state reaction outcomes:
- Chemical and morphological properties of reagents, including their reactivity, surface area, and the free energy change associated with the reaction [1].
- Reaction conditions such as temperature, pressure, and atmospheric environment [1].
- Mixing efficiency, where improved homogeneity of reactants enhances reactivity through a higher surface-volume ratio [91].
The Critical Role of Calcination Parameters
Calcination conditions, particularly temperature and duration, profoundly influence the phase purity, crystallinity, and morphology of the final product. Studies on cobalt antimonate (CoSb₂O₆) synthesis reveal how these parameters directly impact material characteristics:
- Temperature Dependence: The highest yield of CoSb₂O₆ was observed in samples calcined at 600°C for 6 hours, with higher temperatures promoting refractory phases like Co₃O₄ [92].
- Time Dependence: Extended calcination times do not necessarily enhance desired phase formation. For CoSb₂O₆, the optimal calcination time was 6 hours, not 12 hours [92].
- Atmospheric Effects: Reactions conducted under static air conditions prevent control of oxygen content, potentially leading to multiple stable phases in the final product [92].
Table 1: Impact of Calcination Parameters on CoSb₂O₆ Synthesis
| Calcination Temperature (°C) | Calcination Time (hours) | CoSb₂O₆ Content | Secondary Phases Observed | Key Findings |
|---|---|---|---|---|
| 600 | 6 | Highest | Co₃O₄, Sb₂O₃ | Optimal parameters for maximizing target phase |
| 600 | 12 | Increased | Co₃O₄, Sb₂O₃ | Longer time increases yield at this temperature |
| >600 | 6 | Decreased | Co₃O₄ (increased) | Higher temperatures favor refractory phases |
| >600 | 12 | Decreased | Co₃O₄ (increased) | Worst combination for target phase formation |
Standardized Experimental Protocols for Comparative Studies
Solid-State Synthesis Workflow
The following diagram illustrates a standardized workflow for solid-state synthesis, incorporating critical steps that must be documented for fair comparisons.
Detailed Methodologies for Key Experiments
Solid-State Synthesis of CoSb₂O₆-Based Electrodes
Objective: To investigate the effects of calcination time and temperature on the formation and electrocatalytic properties of CoSb₂O₆ for the oxygen evolution reaction (OER) in acidic electrolytes [92].
Protocol:
- Precursor Preparation: Use stoichiometric amounts of cobalt and antimony precursors.
- Mixing: Combine precursors thoroughly using a solvent-free solid-state method.
- Calcination: Heat samples under static air conditions at varying temperatures (e.g., 600°C, 700°C, 800°C) for different durations (e.g., 6 hours vs. 12 hours).
- Characterization:
- Structural: Powder X-ray diffraction (PXRD) to identify crystalline phases and calculate crystallite size.
- Compositional: Energy-dispersive X-ray spectroscopy (EDX) for semiquantitative analysis of phase percentages and atomic ratios.
- Textural: Brunauer-Emmett-Teller (BET) analysis to determine specific surface area, total pore volume, and average pore width.
- Morphological: Field-emission scanning electron microscopy (FESEM) to examine particle morphology and distribution.
- Electrochemical: Measure OER activity including Tafel slopes and overpotentials.
Synthesis of ZrV₂O₇ with Phase Purity Control
Objective: To produce phase-pure negative thermal expansion material ZrV₂O₇ through solid-state reaction and sol-gel methods, comparing the influence of synthesis methods on purity and homogeneity [91].
Protocol:
- Solid-State Route:
- Precursors: Use ZrO₂ and V₂O₅ in stoichiometric ratios.
- Milling: Employ extended milling times (15 min, 40 min, 180 min) to improve homogeneity.
- Calcination: Conduct multiple calcination cycles (1, 2, or 3 times) at 700°C for 5-20 hours with intermediate grinding steps.
- Quenching: Rapidly cool selected samples in air or liquid nitrogen to prevent undesired low-temperature processes.
- Sol-Gel Route:
- Achieve "near-atomic" level mixing of zirconium and vanadium precursors in solution.
- Process through gelation, drying, and calcination stages.
- Characterization:
- Phase Purity: X-ray diffraction (XRD) compared against simulated patterns from optimized crystal structures.
- Structural Analysis: Raman spectroscopy with interpretation based on ab initio simulated phonon data.
- Homogeneity Assessment: Advanced analytical techniques to distinguish subtle structural differences.
Comparative Data Analysis Framework
Quantitative Comparison of Synthesis Outcomes
Table 2: Synthesis Outcome Comparison for Different Materials Systems
| Material System | Optimal Synthesis Parameters | Key Performance Metrics | Impact of Synthesis Variations |
|---|---|---|---|
| CoSb₂O₆ | 600°C, 6 hours | • Tafel slope• Overpotential• Phase purity (%) | Higher temperatures (≥700°C) promote Co₃O₄ formation, increasing overpotential to >1V [92] |
| ZrV₂O₇ | Multiple cycles at 700°C with extended milling | • Phase purity• Negative thermal expansion coefficient• Homogeneity | Inadequate milling leads to ZrO₂ remnants; insufficient cycles yield multiphase ceramics [91] |
| LNMO Hollow Microspheres | Kirkendall-effect driven SSR | • Discharge capacity (mAh/g)• Capacity retention (%) | Hollow structures facilitate Li+ transfer, achieving 115 mAh/g at 5C and 96.6% retention after 200 cycles [1] |
The Scientist's Toolkit: Essential Research Reagents
Table 3: Essential Materials and Equipment for Solid-State Synthesis
| Category | Specific Items | Function in Synthesis |
|---|---|---|
| Precursor Materials | Metal oxides (ZrO₂, V₂O₅), Carbonates, Chloride salts | Provide cation sources for final compound formation [91] |
| Mixing Equipment | Mortar and pestle, Ball mill, High-energy mills | Achieve homogeneous mixing of solid precursors; reduced particle size enhances reactivity [91] |
| Heating Equipment | Programmable muffle furnace, Tube furnace with atmosphere control | Enable controlled calcination at high temperatures (typically 600-1200°C) with atmospheric regulation [92] |
| Characterization Tools | XRD, Raman spectroscopy, SEM/EDX, BET surface area analyzer | Verify phase purity, morphology, composition, and textural properties [92] [91] |
Advanced Framework for Experimental Design
Goal-Oriented Experimental Design
Modern experimental design should align with specific research goals rather than aiming for comprehensive model learning. The Goal-Oriented Causal Bayesian Experimental Design (GO-CBED) framework provides a structured approach for selecting interventions that maximize expected information gain (EIG) on user-specified causal quantities of interest [93]. This is particularly valuable in solid-state synthesis where experimental resources are limited and the parameter space is vast.
Parameter Sensitivity-Driven Clustering
The PARSEC (PARameter SEnsitivity Clustering) framework offers a systematic approach for designing informative experiments through parameter sensitivity analysis [94]. This method:
- Computes parameter sensitivity indices (PSI) for measurable variables
- Identifies measurement combinations with distinct PSI vector representations
- Clusters PSIs based on Euclidean distance to identify minimal measurement sets
- Captures essential system dynamics for precise parameter estimation
The following diagram illustrates the PARSEC framework workflow for rationalized experiment design.
Establishing a framework for fair comparison of materials requires meticulous documentation and standardization of synthesis conditions. Key parameters including precursor materials, milling duration, calcination temperature and time, atmospheric conditions, and intermediate processing steps must be systematically reported. The experimental frameworks outlined in this guide enable researchers to conduct meaningful comparisons by controlling for synthesis-related variability, thereby advancing the field of solid-state chemistry through more reproducible and comparable research outcomes.
Future directions should emphasize the adoption of goal-oriented experimental design principles and sensitivity-based clustering approaches to optimize synthesis parameter selection, ultimately accelerating materials discovery and development through more efficient and targeted experimentation.
Conclusion
The choice between direct solid-state and flux-assisted synthesis is pivotal, directly influencing critical material properties such as crystallinity, defect density, and ultimately, functional performance in applications from photocatalysis to energy storage. Flux methods demonstrate clear advantages in reducing reaction temperatures, shortening synthesis times, and improving particle morphology, though they introduce complexity in post-synthesis purification. Future research should focus on developing novel, low-melting-point flux reagents, optimizing reactor designs for large-scale production, and deepening the understanding of reaction mechanisms at the molecular level. Embracing these advanced synthetic strategies will accelerate the development of next-generation materials for biomedical and clinical research, enabling more efficient and scalable production of high-performance compounds.
References
- 1. Solid State Reaction - an overview [https://www.sciencedirect.com/topics/chemistry/solid-state-reaction]
- 2. How to accelerate the inorganic materials synthesis [https://pmc.ncbi.nlm.nih.gov/articles/PMC11960098/]
- 3. Mn 4+ Phosphor Prepared by Solid-State Reaction ... [https://www.mdpi.com/2673-6497/6/3/53]
- 4. Autonomous and dynamic precursor selection for solid- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10618174/]
- 5. Solid-state synthesizability predictions using positive ... [https://pubs.rsc.org/en/content/articlehtml/2025/dd/d5dd00065c]
- 6. Reproducible high-quality perovskite single crystals by flux ... [https://www.nature.com/articles/s44160-024-00576-8]
- 7. Molten-salt assisted synthesis of two-dimensional materials ... [https://www.sciencedirect.com/science/article/abs/pii/S2468519423000460]
- 8. Flux Method - an overview [https://www.sciencedirect.com/topics/materials-science/flux-method]
- 9. Synthesis of a series of rare-earth-based multi-anion ... [https://pubs.rsc.org/en/content/articlehtml/2025/dt/d4dt03506b]
- 10. Flux-assisted synthesis of tungsten-doped layered ... [https://pubs.rsc.org/en/content/articlelanding/2023/cc/d2cc05806e]
- 11. Novel flux-assisted synthesis for enhanced afterglow ... [https://www.sciencedirect.com/science/article/abs/pii/S0925838816342451]
- 12. Diamond-Like HgIn2Q4 (Q = Se, Te) Synthesized by Flux- ... [https://www.nature.com/nature-index/article/10.1021/acs.inorgchem.5c02378]
- 13. Emerging processing guidelines for solid electrolytes in the ... [https://pubs.rsc.org/en/content/articlehtml/2025/cs/d5cs00358j]
- 14. Mass transport effects during measurements of gas–solid ... [https://www.sciencedirect.com/science/article/abs/pii/S000925090600666X]
- 15. Understanding of Solid-Fluid Kinetics and Mass Transfer [https://www.frontiersin.org/journals/chemical-engineering/articles/10.3389/fceng.2020.00006/full]
- 16. Optimized Flux Single-Crystal Growth of the Quantum Spin ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12186254/]
- 17. Comparative investigation on carbon dots and chloride ... [https://www.sciencedirect.com/science/article/abs/pii/S0025540824004987]
- 18. Quantifying the regime of thermodynamic control for solid- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11221506/]
- 19. A Comparative Study of Sol–Gel and Solid-State Methods ... [https://www.mdpi.com/2504-477X/9/11/632]
- 20. Flux Method - an overview [https://www.sciencedirect.com/topics/physics-and-astronomy/flux-method]
- 21. Text-mined dataset of inorganic materials synthesis recipes [https://pmc.ncbi.nlm.nih.gov/articles/PMC6794279/]
- 22. Solid-state vs. spray-drying synthesis for Mg-doped P2–Na ... [https://pubs.rsc.org/en/content/articlehtml/2025/ta/d5ta04988a]
- 23. Solid-State Reaction Route - an overview [https://www.sciencedirect.com/topics/materials-science/solid-state-reaction-route]
- 24. Solid-state reaction route [https://en.wikipedia.org/wiki/Solid-state_reaction_route]
- 25. Autonomous and dynamic precursor selection for solid- ... [https://www.nature.com/articles/s41467-023-42329-9]
- 26. Overlooked impact of precursor mixing: Implications in the ... [https://www.sciencedirect.com/science/article/abs/pii/S2095495623001912]
- 27. Effect of calcination on the structural, morphological and ... [https://www.sciencedirect.com/science/article/abs/pii/S0022459624004237]
- 28. Flux synthesis, structure and physical properties of new ... [https://www.sciencedirect.com/science/article/abs/pii/S0925838800014110]
- 29. Solid-State Reaction Heterogeneity During Calcination of ... [https://pubmed.ncbi.nlm.nih.gov/36583605/]
- 30. Flux growth of high entropy layered perovskite-type oxysulfide ... [https://pubs.rsc.org/en/Content/ArticleHtml/2025/CE/d5ce00631g]
- 31. CaCl versus MagCl - Calcium Chloride [https://www.akjchem.com/cacl-versus-magcl]
- 32. Photocatalytic performance of Y2Ti2O5S2 prepared via ... [https://pubs.rsc.org/en/content/articlelanding/2025/ta/d4ta08167f]
- 33. Photocatalytic performance of Y 2 Ti 2 O 5 S 2 prepared via ... [https://pubs.rsc.org/en/content/articlehtml/2025/ta/d4ta08167f]
- 34. Flux-Assisted Synthesis of Y2 Ti2 O5 S2 for Photocatalytic ... [https://pubmed.ncbi.nlm.nih.gov/37653542/]
- 35. Synthesis, characterization, and preliminary insights of ... [https://www.nature.com/articles/s41598-023-46960-w]
- 36. Spinel ferrite (AFe2O4)-based heterostructured designs for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9056412/]
- 37. A comparative study of spinel ZnFe 2 O 4 ferrites obtained ... [https://www.sciencedirect.com/science/article/abs/pii/S0272884221004259]
- 38. Synthesis of ZnFe 2 O 4 nanoparticles via a modified ... [https://www.sciencedirect.com/science/article/abs/pii/S1044580325006382]
- 39. Synthesis and characterization of ZnFe2O4 nanoparticles ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4632970/]
- 40. Comparison of closed and open thermochemical ... [https://www.sciencedirect.com/science/article/abs/pii/S0360544214006641]
- 41. Prototype thermochemical heat storage with open reactor ... [https://www.sciencedirect.com/science/article/abs/pii/S0306261913001013]
- 42. Flux growth of yttrium calcium oxy borate (YCOB) single ... [https://www.sciencedirect.com/science/article/abs/pii/S0022024808010208]
- 43. Small Modular Reactors: 'Sealing' the Deal [https://www.powermag.com/small-modular-reactors-sealing-the-deal/]
- 44. Grand challenges in advanced nuclear reactor design [https://www.frontiersin.org/journals/nuclear-engineering/articles/10.3389/fnuen.2022.1000754/full]
- 45. Fluxing Process in Metal Melting: How It Works, Types, & ... [https://www.okonrecycling.com/industrial-scrap-metal-recycling/steel-and-aluminum/fluxing-process-metal-melting/]
- 46. Synthesis of Er 2 Ir 2 O 7 pyrochlore iridate by solid-state- ... [https://www.sciencedirect.com/science/article/abs/pii/S025405842031227X]
- 47. Raw materials for growing single crystals based on YAG [https://link.springer.com/article/10.1007/s10854-025-15882-w]
- 48. Crystallization and particle size distribution of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11845732/]
- 49. Predictive screening of phase stability in high-entropy ceramics [https://pubs.rsc.org/en/content/articlehtml/2025/ma/d5ma00079c]
- 50. Probing solid-state diffusion and its synergistic role with ... [https://www.sciopen.com/article/10.26599/JAC.2025.9221078]
- 51. ReactCA: A Cellular Automaton for Predicting Phase ... [https://arxiv.org/html/2407.19124v1]
- 52. Flux Growth of Phosphide and Arsenide Crystals - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC7142258/]
- 53. Modelling and simulation of the flux responses of a gas– ... [https://www.sciencedirect.com/science/article/abs/pii/S0009250901004365]
- 54. Achieving positive temperature-dependent thermal ... [https://www.sciopen.com/article/10.26599/JAC.2025.9221183]
- 55. Determination of the prospects for the use ... [https://www.sciencedirect.com/science/article/pii/S2666539525001051]
- 56. Limitation of solid-reaction technique in a controlled ... [https://www.sciencedirect.com/science/article/pii/S2307410823000317]
- 57. Oligonucleotide Purification Methods for Your Research [https://www.thermofisher.com/us/en/home/brands/thermo-scientific/molecular-biology/molecular-biology-learning-center/molecular-biology-resource-library/spotlight-articles/oligonucleotide-purification-methods.html]
- 58. Oligo purification methods: which type should I choose? | IDT [https://www.idtdna.com/page/support-and-education/decoded-plus/which-type-of-purification-should-i-choose/]
- 59. Vacancies in sulfides facilitate fluid-induced solid-state ... [https://www.nature.com/articles/s41467-025-57171-4]
- 60. Oligonucleotide synthesis basics: Key takeaways for post ... [https://blog.biosearchtech.com/oligonucleotide-synthesis-basics-key-takeaways-for-post-synthesis-processing]
- 61. Impact of mechanical activation and mechanochemical ... [https://www.sciencedirect.com/science/article/abs/pii/S0892687524003911]
- 62. Mechanochemical activation of natural metal sulfide ... [https://www.sciencedirect.com/science/article/abs/pii/S1383586623016180]
- 63. Scaling theory for the kinetics of mechanochemical reactions ... [https://pubs.rsc.org/fr/content/articlehtml/2025/mr/d4mr00091a]
- 64. Sulfurizing Reagent II and its use in Synthesizing ... [https://www.glenresearch.com/reports/gr18-18]
- 65. Top 5 Uses of Sulfur Monochloride in 2025 | Integration Note [https://www.linkedin.com/pulse/sulfur-monochloride-real-world-5-uses-youll-actually-see-mqtef]
- 66. Scientists develop new method for creating promising ... [https://www.sciencedaily.com/releases/2022/02/220214121251.htm]
- 67. Sulfurization - an overview | ScienceDirect Topics [https://www.sciencedirect.com/topics/chemistry/sulfurization]
- 68. X-ray diffraction (XRD) basics and application [https://chem.libretexts.org/Courses/Franklin_and_Marshall_College/Introduction_to_Materials_Characterization__CHM_412_Collaborative_Text/Diffraction_Techniques/X-ray_diffraction_(XRD)_basics_and_application]
- 69. X-ray Diffraction Techniques for Mineral Characterization [https://www.mdpi.com/2075-163X/12/2/205]
- 70. XRD | West Campus Materials Characterization Core [https://ywcmatsci.yale.edu/xrd]
- 71. Scanning Electron Microscopy [https://www.nanoscience.com/techniques/scanning-electron-microscopy/]
- 72. Scanning Electron Microscopy Characterization & Testing [https://www.innovatechlabs.com/materials-analysis-electron-microscopy/]
- 73. Basics of Electrochemical Impedance Spectroscopy [https://www.gamry.com/application-notes/EIS/basics-of-electrochemical-impedance-spectroscopy/]
- 74. Thermal Analysis Techniques for Material Characterization [https://measurlabs.com/blog/thermal-analysis-technique-comparison/]
- 75. XRD and combined SEM-EDS analysis of long-term ... [https://www.sciencedirect.com/science/article/abs/pii/S0254058421011561]
- 76. Asymmetric coordination enhances the synergy of Pt ... [https://www.nature.com/articles/s41467-025-63637-2]
- 77. Experimental and theoretical insights into LDH based on ... [https://www.nature.com/articles/s41598-025-17648-0]
- 78. Photocatalytic Hydrogen Production Performance of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12471888/]
- 79. Recent Progress and Future Perspectives of MNb2O6 ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12347838/]
- 80. Photocatalytic hydrogen evolution: Recent advances in ... [https://www.sciencedirect.com/science/article/abs/pii/S0360319925011838]
- 81. Comparative insights into CuS and Bi 2 S 3 -based ... [https://link.springer.com/article/10.1007/s42452-025-07600-2]
- 82. Top 179 Energy storage papers published in 2025 [https://scispace.com/journals/energy-storage-2fnn1xbw/2025]
- 83. Key Performance Metrics of ESS | Cycle Life & Efficiency [https://ffdpower.com/key-performance-metrics-of-ess/]
- 84. Comprehensive Guide to Key Performance Indicators of ... [https://www.tls-containers.com/tls-blog/comprehensive-guide-to-key-performance-indicators-of-energy-storage-systems]
- 85. and Calcium-Ion Conductors Via Solid-State Metathesis ... [https://chemrxiv.org/engage/chemrxiv/article-details/6917ce14a10c9f5ca17efdc0]
- 86. Crystallinity analysis [https://rigaku.com/resources/techniques/crystallinity-analysis]
- 87. Determination of % Crystallinity in Polymers [https://mcl.mse.utah.edu/determination-of-crystallinity-in-polymers/]
- 88. Toward high-throughput defect density quantification [https://pubmed.ncbi.nlm.nih.gov/31419611/]
- 89. Advancements and Challenges in Organic–Inorganic Composite Solid Electrolytes for All-Solid-State Lithium Batteries | Nano-Micro Letters [https://link.springer.com/article/10.1007/s40820-024-01498-y]
- 90. Defect Density - an overview [https://www.sciencedirect.com/topics/materials-science/defect-density]
- 91. Synthesis and phase purity of the negative thermal expansion ... [https://pubs.rsc.org/en/content/articlehtml/2025/tc/d4tc04095c]
- 92. Solid-State Reaction Synthesis of CoSb 2 O 6 -Based ... [https://www.mdpi.com/2073-4344/15/1/68]
- 93. Goal-Oriented Sequential Bayesian Experimental Design ... [https://arxiv.org/html/2507.07359v1]
- 94. Rationalised experiment design for parameter estimation ... [https://www.nature.com/articles/s41598-024-75539-2]